Ⅰ. Introduction
Inadequate alveolar ridge volume often leads to improper three-dimensional implant positioning, adversely affecting long-term stability and esthetic outcomes.1,2Therefore, horizontal ridge augmentation (HRA) is often essential prior to or in conjunction with implant placement in anatomically unfavorable ridges.
Although guided bone regeneration (GBR) and block bone grafting are commonly used techniques in the posterior mandible, the unfavorable residual ridge morphology in this region can limit the effectiveness of conventional GBR.3,4Furthermore, autogenous block bone grafts are associated with disadvantages such as donor site morbidity, challenges in soft tissue management, and the requirement for graft fixation.5,6
Recombinant human bone morphogenetic protein-2 (rhBMP-2) is a potent osteoinductive factor that promotes osteoblast proliferation, osteogenic differentiation, and mesenchymal cell recruitment.7,8 Owing to its low molecular weight and high solubility, rhBMP-2 readily diffuses into body fluids; therefore, employing a suitable carrier matrix is imperative.9,10
Acellular dermal matrix (ADM), originally developed as a dermal substitute for skin grafting, has been adapted for dental applications as a substitute for autogenous connective tissue grafts and as a barrier membrane in periodontal regenerative procedures. ADM offers several advantages including high tensile strength, excellent clinical workability, resistance to infection, and the promotion of neovascularization owing to its high collagen content.11 These properties suggest that ADM may be a promising carrier for rhBMP-2 in bone regeneration.
This report presents two clinical cases in which rhBMP-2-loaded ADM was applied to patients with flat and steep buccal alveolar slopes in the posterior mandible, with the primary aim of achieving horizontal bone augmentation. Clinical and radiographic outcomes, as well as subsequent functional loading of the implants were evaluated.
Ⅱ. Case Report
1. Case 1
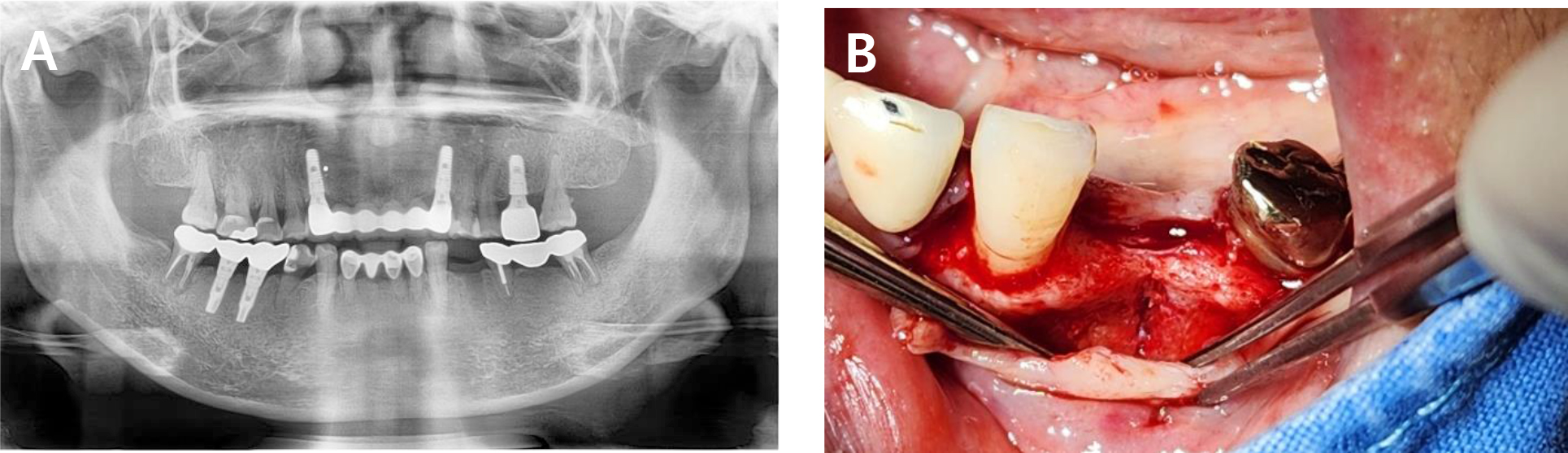
A 63-year-old female with no significant systemic medical history visited our clinic for implant placement at the mandibular left first premolar, which had been extracted 4 months earlier due to dental caries. Preoperative clinical and radiographic examination revealed a flat residual ridge with a steep buccal slope. Ideal prosthetically driven implant placement resulted in buccal thread exposure, necessitating simultaneous HRA (Fig. 1).
To compensate for the anticipated horizontal deficiency of approximately 1.5 mm, bone grafting was planned using a combination of dehydrated ADM (MegaDerm® thickness: 0.3–0.6 mm; L & C Bio, Seoul, Korea) loaded with rhBMP-2 (Novosis®; CGbio, Seoul, Korea).
Under local anesthesia with 2% lidocaine, a crestal incision was made and a full-thickness mucoperiosteal flap was elevated. A 3.5 mm diameter implant was placed according to prosthetic considerations, followed by cover screw installation. As expected, the buccal threads were exposed immediately after placement. The ADM was trimmed to fully cover all areas where buccal bone thickness was clinically less than 1.5 mm, and then immersed in 0.25 mg rhBMP-2 dissolved in 0.5 mL of normal saline for 1 min. The soaked ADM was folded once, directly applied to the defect site, and secured with simple interrupted sutures (Fig. 2).
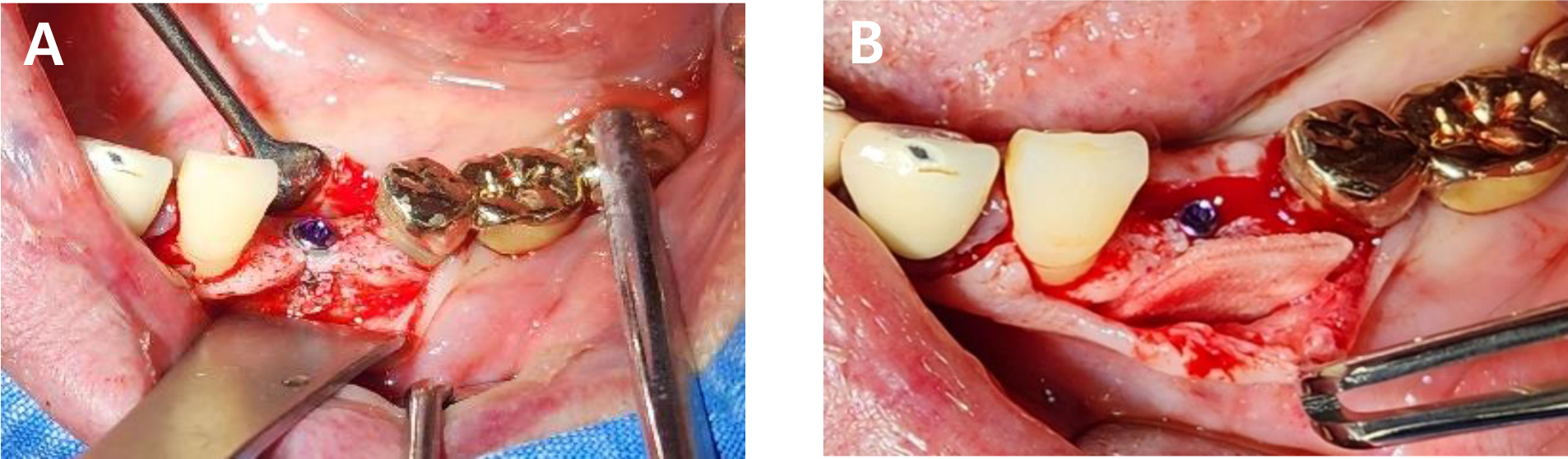
At the 6-month follow-up, the patient returned for second-stage surgery. Flap reflection revealed that the area previously covered by the ADM had been replaced with newly formed bone. Approximately 1.5 mm of horizontal bone gain was observed, and the previously exposed buccal implant threads were now completely covered by new bone (Fig. 3).
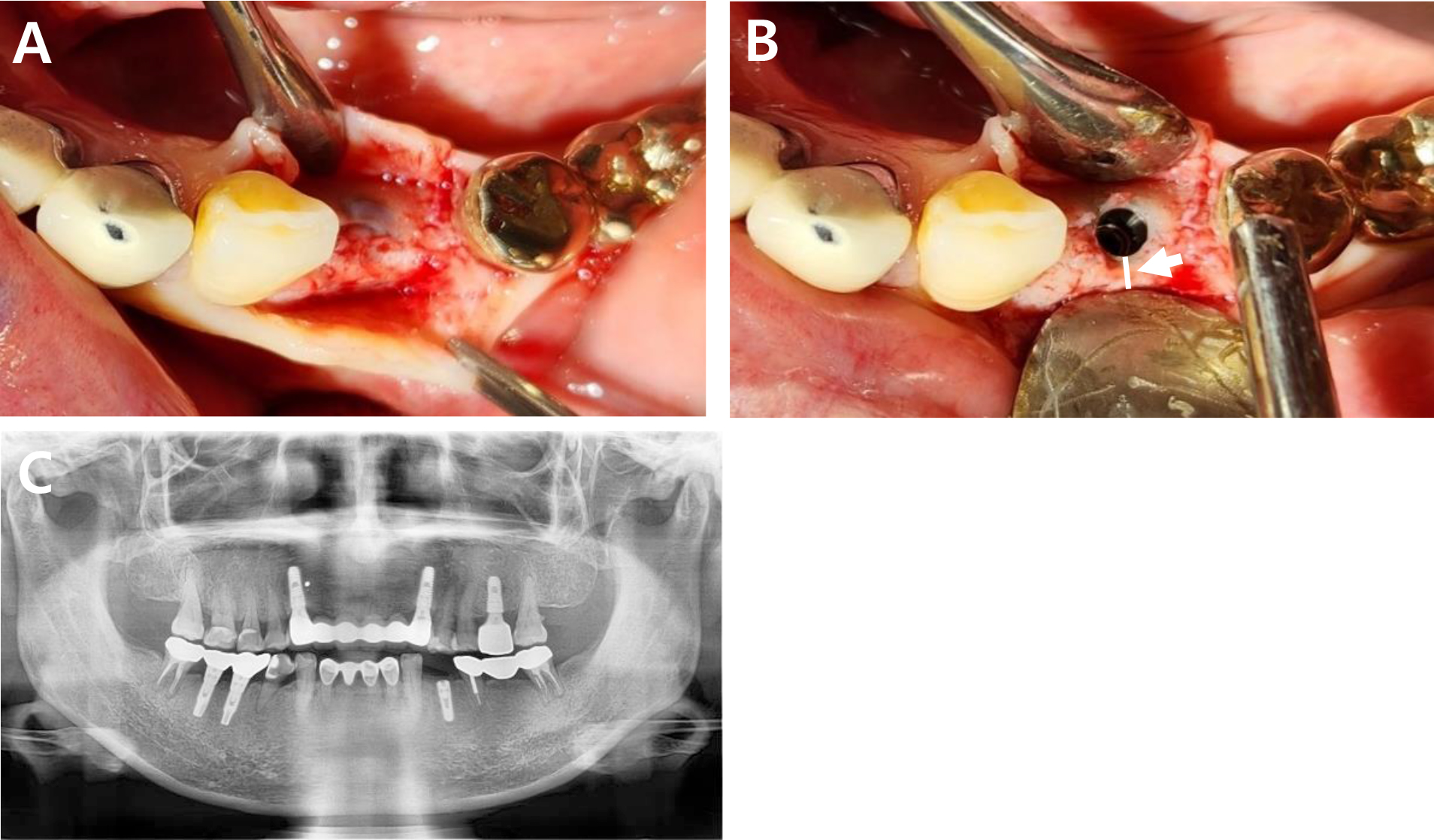
Fig. 3
(A) Clinical photograph showing horizontally augmented alveolar ridge six months after implant placement. (B) Clinical photograph after removal of the cover screw. (C) Six-month postoperative panoramic radiograph. Showing the newly formed bone (white arrow), measuring approximately 1.5 mm in width.
No signs of peri-implantitis or soft tissue inflammation were observed during the 13-month follow-up period after prosthetic loading.
2. Case 2
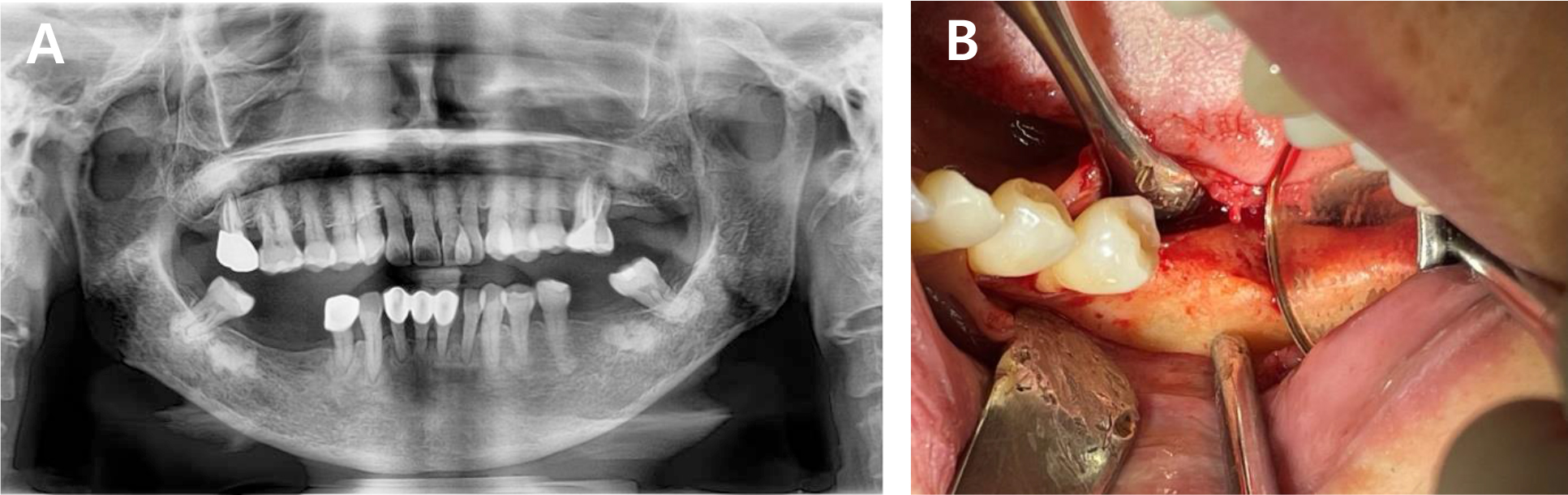
A 57-year-old female undergoing continuous ambulatory peritoneal dialysis at our hospital for end-stage renal disease visited our clinic for implant placement at the mandibular left first molar site, which had been lost due to chronic periodontitis. As alveolar bone loss progressed, the left mandibular second molar was considered non-restorable. As a result, the second molar was scheduled for extraction, and an implant was planned for the first molar site (Fig. 4).
Preoperative cone-beam computed tomography (CBCT) revealed horizontal alveolar bone deficiency due to long-term edentulism. To ensure long-term implant success, approximately 1–2 mm of HRA was considered necessary. Accordingly, simultaneous placement of a 4 mm diameter implant was planned, along with HRA using ADM loaded with rhBMP-2.
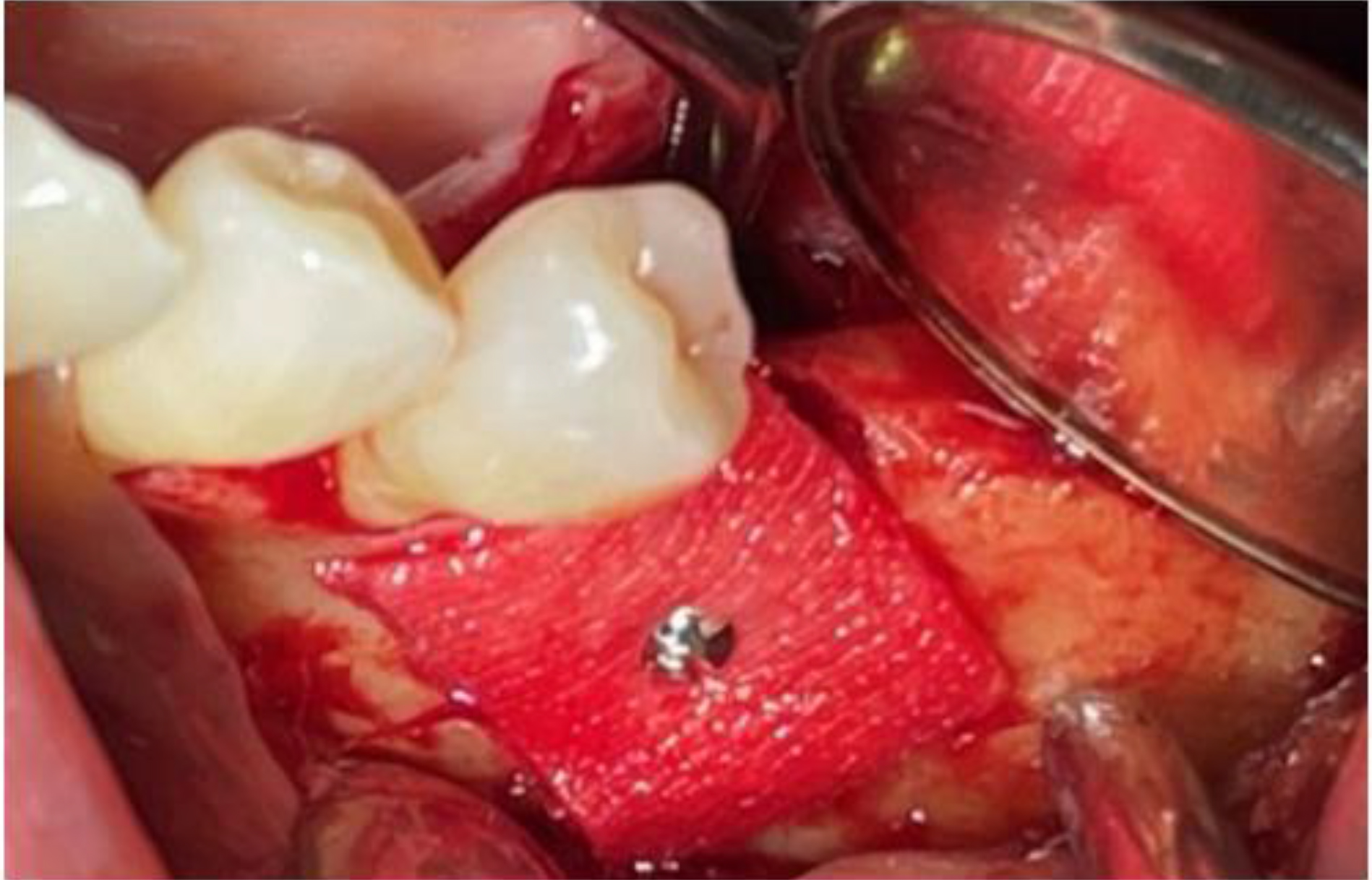
During surgery, the dehydrated ADM was carefully cut and folded to achieve a total thickness of approximately 0.6–1.2 mm. It was then immersed in 0.25 mg of rhBMP-2 dissolved in 0.5 mL of normal saline for 1 min. The prepared ADM was applied to the buccal bone defect and a titanium fixation screw was used to stabilize the graft material in position (Fig. 5). The surgical site was then carefully closed using simple interrupted sutures.
CBCT analysis performed 5 months postoperatively confirmed a horizontal bone gain of approximately 1.7 mm. Newly formed bone with homogeneous density and continuity was observed around the implant (Fig. 6). Throughout the 10-month functional loading period, no signs of inflammation, bone resorption, or other complications were observed in the surrounding hard or soft tissues. The implant remained functionally stable.
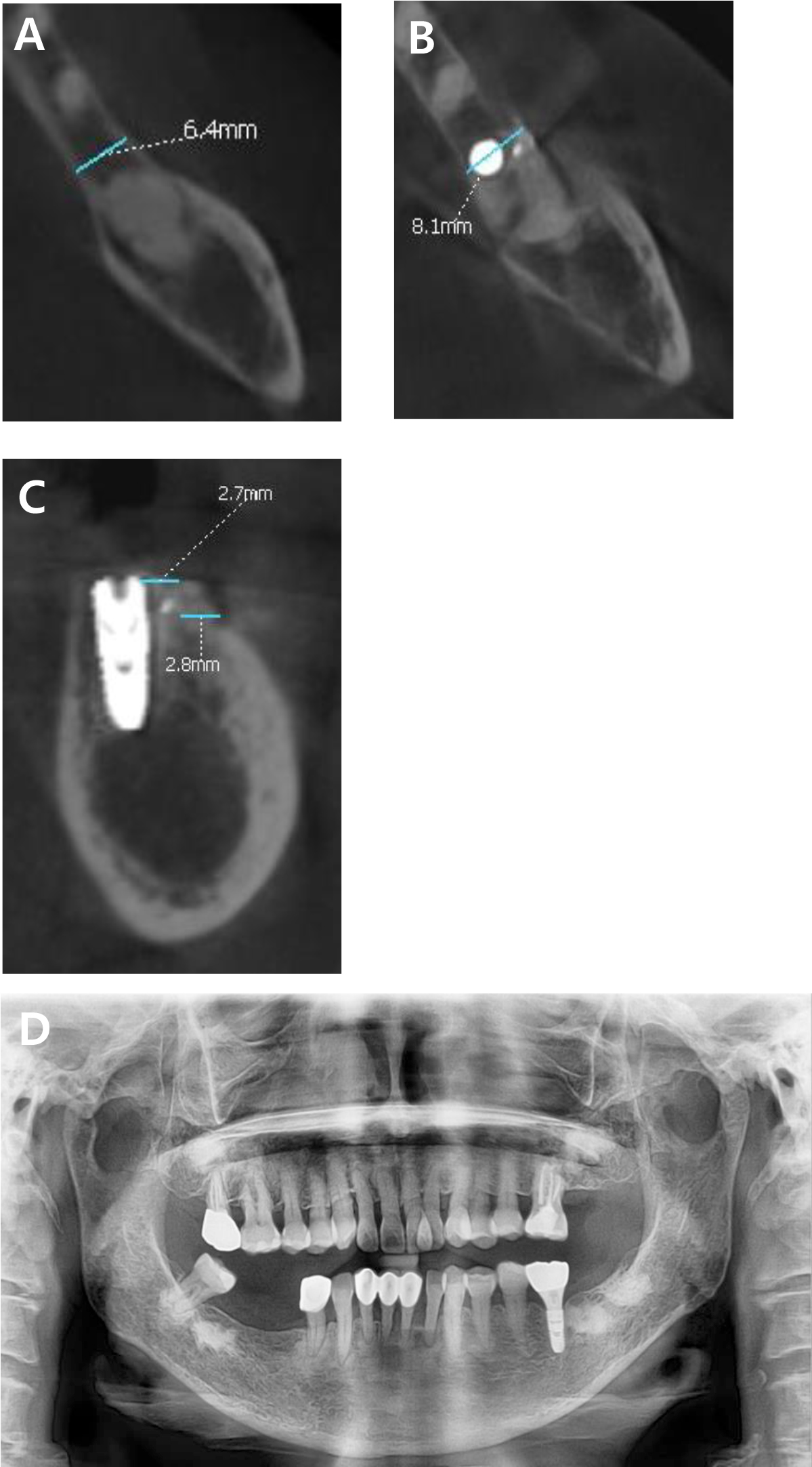
Fig. 6
(A) Preoperative cone-beam computed tomography (CBCT) scan showing the alveolar bone width at the level corresponding to the fixation screw position in the postoperative scan. (B) CBCT scan 5 months postoperatively showing a horizontal bone gain of 1.7 mm. (C) Sagittal CBCT view showing buccal cortical bone thickness at the level of the bone crest and 2 mm below the crest after surgery. (D) Panoramic radiograph after functional loading.
Ⅲ. Discussion
ADM has been widely utilized as a scaffold material in various reconstructive procedures because of its high collagen content and mechanical stability.12,13 More recently, its applications have expanded into the dental field, including periodontal plastic surgery and bone regeneration techniques.14,15 Unlike conventional collagen sponges or synthetic materials such as β-tricalcium phosphate, ADM provides a biologically active matrix that supports cell adhesion, migration, neovascularization, and soft tissue integration.16,17,18
These properties make ADM a promising carrier for rhBMP-2, potentially enhancing bone regeneration. The combination of ADM and rhBMP-2 has demonstrated osteoinductive potential in animal models. Previous animal studies demonstrated partial replacement of ADM by dense collagen bundles and elastic fibers, accompanied by progressive new bone formation.19 However, clinical studies evaluating the use of this combination in humans—particularly under functional loading conditions—remain limited. In the present cases, ADM effectively served as both a delivery system for rhBMP-2 and as a structural scaffold that maintained space at the defect site, thereby facilitating predictable bone regeneration.
A clinically significant consideration is whether the regenerated bone can support long-term functional loading of the implant. In this study, both CBCT and clinical evaluations confirmed that the newly formed bone was continuous with the native alveolar ridge. Furthermore, stable peri-implant conditions were observed without any signs of inflammation or bone loss during follow-up periods of 13 and 10 months, respectively. These findings strongly suggest that ADM-based ridge augmentation may contribute not only to short-term bone formation but also to sustained long-term implant survival and stability.
Nevertheless, a limitation of this study is the absence of histological evaluation, which restricts deeper assessment of the biological characteristics of the regenerated bone. In addition, long-term follow-up periods are necessary to further verify the sustained stability of the augmented sites.
Despite these limitations, this case report demonstrated the clinical feasibility of using rhBMP-2-loaded ADM for horizontal ridge augmentation in humans. This method yielded favorable bone regeneration outcomes and maintained stable implant conditions under short-term functional loading. Further prospective, controlled studies, including histological analyses, are warranted to validate the broader applicability of this approach in implant dentistry.
Ⅳ. Conclusion
This case report demonstrates that ADM loaded with rhBMP-2 can effectively induce predictable horizontal bone augmentation in the posterior mandibular region without the need for additional grafting materials or barrier membranes. The regenerated bone provided stable support for functional implant loading. Owing to its ease of handling and the ability to achieve tension-free primary closure without additional procedures, this technique offers both clinical efficiency and procedural safety in cases requiring limited augmentation. Further prospective studies with long-term follow-up are warranted to further validate and expand the clinical applicability of this approach.