Ⅰ. Introduction
Medication-related osteonecrosis of the jaw (MRONJ) is a severe complication associated with antiresorptive therapy, and its relationship with dental implants remains controversial. Recent studies have reported that implant placement does not increase the incidence of MRONJ in patients with osteoporosis or cancer.1, 2 However, other reports have documented MRONJ development either following implant surgery or around previously osseointegrated implants.3, 4, 5
In the literature, the term ‘implant surgery-triggered MRONJ’ refers to cases in which MRONJ develops in association with implant surgery, whereas ‘implant presence-triggered MRONJ’ denotes cases that occur after complete osseointegration.4 However, some experts argue that these terms may not be entirely appropriate, as a definitive cause-and-effect relationship has not been established, and scientific evidence remains limited.2 As noted above, the mechanisms underlying implant-related MRONJ remain unclear, and multiple contributing factors have been suggested.
Souza et al. reported a significant exacerbation of inflammation and an increased amount of nonvital bone tissue surrounding implants in the presence of peri-implantitis.6 Functional loading of implants can also induce microdamage and potentially heighten MRONJ susceptibility.7 Additionally, long-term bisphosphonate therapy or systemic corticosteroid administration has been identified as a risk factor for MRONJ following implant placement.8, 9
Although the incidence of implant-associated MRONJ remains low, multiple cases have been reported.10 A recent position paper identified delayed MRONJ and implant placement as potential risk factors; however, establishing clear clinical guidelines remains challenging.10, 11 This study presents two cases of MRONJ associated with dental implants and discusses patient management strategies and preventive measures based on a review of the relevant literature.
Ⅱ. Report of Cases
This study adhered to the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Gangnam Severance Hospital (approval No. #3-2025-0091).
1. Case 1
A 68-year-old woman who had been taking Fosamax (alendronate sodium 70 mg with cholecalciferol 5600 IU, orally once weekly) for osteoporosis from January 6, 2014, to November 27, 2020, presented to the Department of Periodontology with discomfort in the mandibular left second molar region (tooth #37). She reported initiating antiresorptive therapy 2 years earlier and, according to her account, had undergone implant placement in the #36 and #37 regions 1 year before her visit.
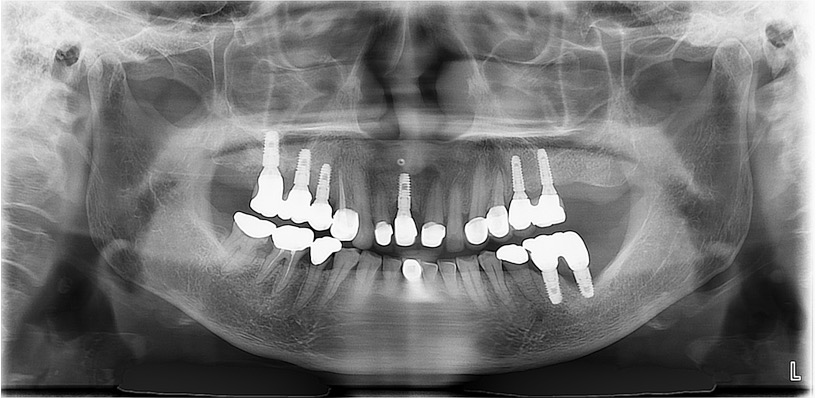
The patient developed persistent pain following osseointegration and prosthetic restoration. A panoramic radiograph revealed no significant lesions except for bone loss on the mesial side of implant #37 (Fig. 1). Despite multiple flap surgeries, the symptoms persisted. On November 26, 2015, implant #37 was removed in the Department of Periodontology at our hospital (Fig. 2A). As discomfort continued, she was referred to the Department of Oral and Maxillofacial Surgery on December 3, 2015, for further evaluation and management. Conservative treatment was attempted for 3 months; however, the lesion progressed, necessitating the extraction of implants #36 and #35 on March 4, 2016 (Fig. 2B and 2C). Six months later, sequestrum formation was observed (Fig. 2D). A sequestrectomy was subsequently performed, resulting in satisfactory healing of the left mandible (Fig. 2E and 2F). The patient discontinued Fosamax and was transitioned to Prolia (denosumab 60 mg, subcutaneous injection every 6 months) on June 2, 2021. Since then, she has continued to receive regular Prolia injections.
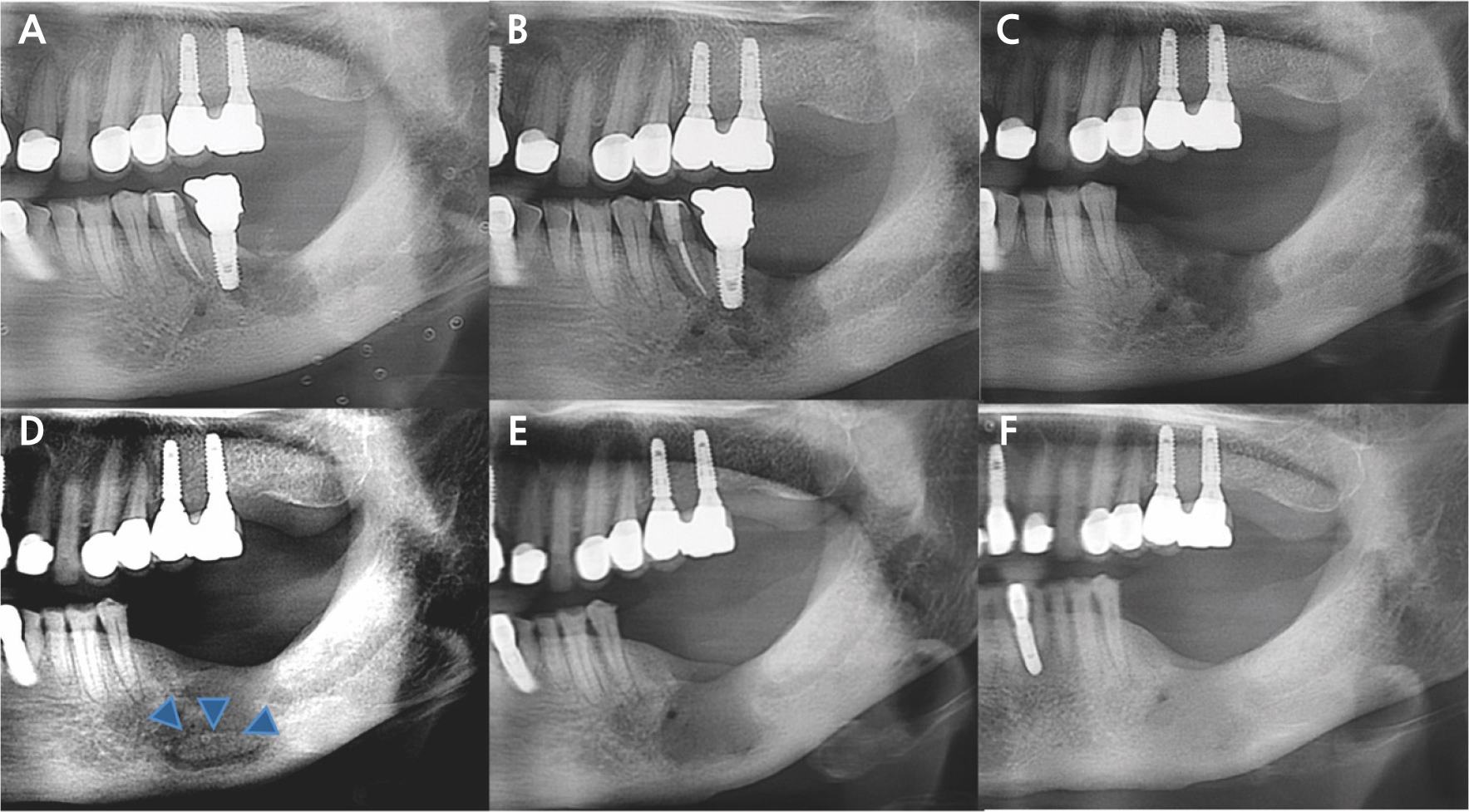
Fig. 2
(A) Implant #37 was removed in the Department of Periodontology, (B, C) The lesion worsened during 3 months of conservative treatment, leading to the extraction of implants #36 and #35, (D) Sequestrum formation was observed after 6 months (blue arrowhead), (E, F) Sequestrectomy was subsequently performed, resulting in satisfactory healing of the left mandible.
According to the 2022 American Association of Oral and Maxillofacial Surgeons (AAOMS) staging system, this case was initially classified as Stage 0 at diagnosis due to nonspecific radiographic findings and persistent pain without bone exposure. The condition later progressed to Stage 2 before surgery, characterized by exposure of necrotic bone accompanied by infection.
2. Case 2
A 72-year-old woman was referred from the Department of Otorhinolaryngology for evaluation and removal of an implant on the right side of the mandible. She reported pricking pain in the right lower lip and mandibular region. Wide excision was planned due to oral mucosal dysplasia involving the lower right molar and tongue areas. Dental history revealed that implants had been placed in #44, #45, and #46 regions 4 years ago. The prosthesis was removed 2 years before because of persistent pain and discomfort. The patient had a history of Bonviva (ibandronate 150 mg with cholecalciferol 24,000 IU, orally once monthly) administration for osteoporosis, taken from 3 years before to 1 month before admission. She had also been treated for spinal stenosis and rheumatoid arthritis with methotrexate and systemic corticosteroids.
A panoramic radiograph obtained at the initial visit revealed sequestrum formation around implants #44, #45, and #46 (Fig. 3A). The patient subsequently underwent sequestrectomy and implant removal. During follow-up, additional sequestrum formation was observed, thereby necessitating a second surgical intervention (Fig. 3B).
Regarding osteoporosis management, the patient received Forsteo (teriparatide, subcutaneous injection) in March and April 2021, followed by Evenity (romosozumab, subcutaneous injection once monthly) for a total of 11 doses between May 2021 and August 2022, with a treatment interruption from May to October 2021. A single Prolia (denosumab, 60 mg subcutaneous injection) dose was administered on September 27, 2022, following completion of Evenity therapy. Subsequently, she was prescribed DicaMAX D (calcium carbonate and cholecalciferol concentrate; Darim Biotech Co., Ltd.) and resumed Forsteo (teriparatide, subcutaneous injection) from January 2024 to December 2024. After sequestrectomy, the right mandibular bone demonstrated satisfactory healing. However, new lesions later developed around the implants in the left maxilla and mandible, extending beyond the alveolar bone (Stage 3), which required additional surgical management (Fig. 3C and 3D). The two cases are summarized chronologically below (Table 1).
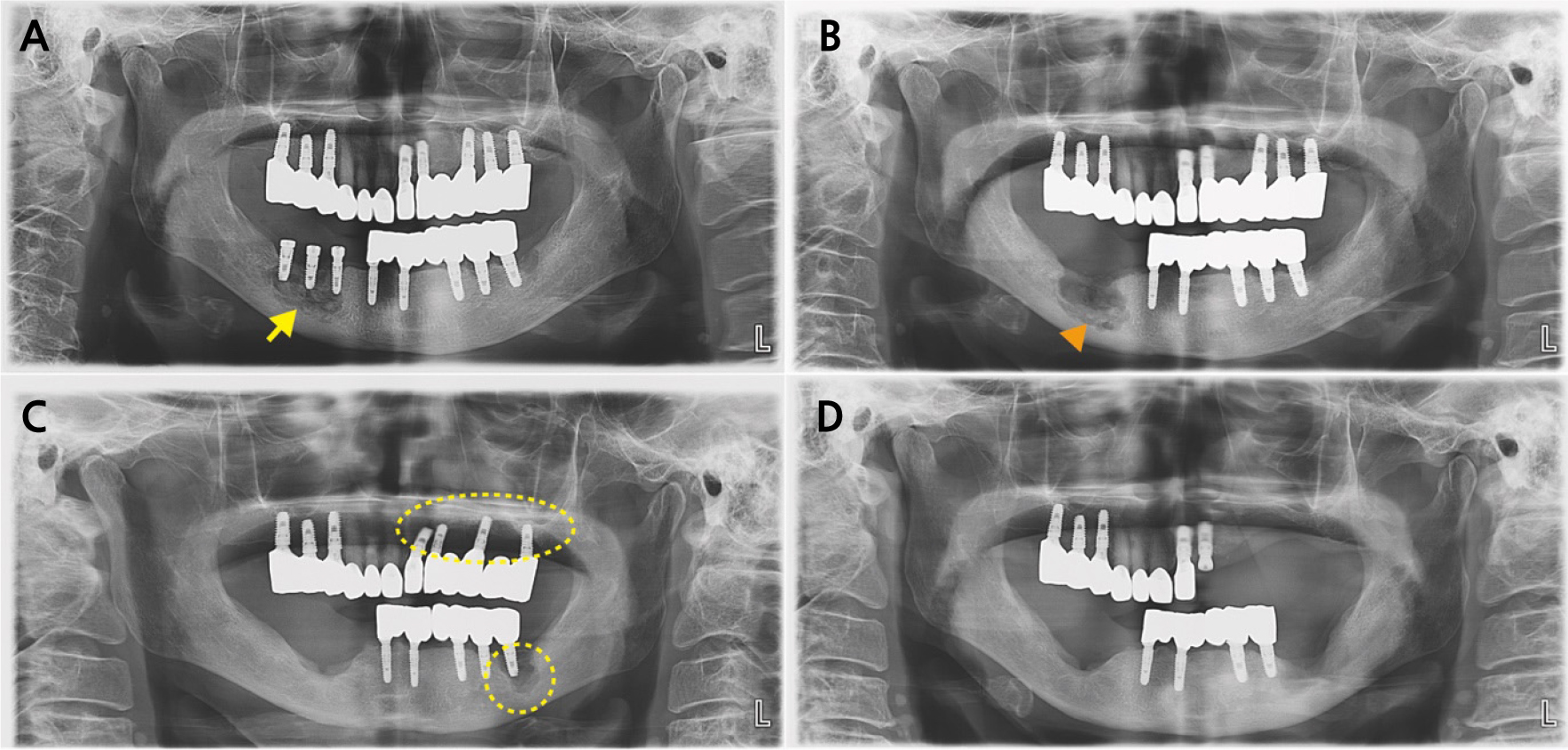
Fig. 3
(A) Panoramic radiograph at the first visit (Case 2) showing sequestrum formation around the implants on #44–#46 (yellow arrow), (B) Additional sequestrum formation was observed during follow-up (orange arrowhead), (C) After sequestrectomy, the right mandible demonstrated satisfactory healing; however, new lesions developed around the implants in the left maxilla and mandible (yellow dotted line), (D) The patient showed a favorable prognosis following additional surgery.
Table 1.
Chronological summary of implant sites (year), antiresorptive medications (dose, route, duration), clinical signs and symptoms (time), treatments, and final outcomes
| Case |
Implant site (year) |
Antiresorptive medication (dose, route, duration) |
Sign and symptom (time) | Treatment | Final treatment outcome |
| 1 | #36, #37 (2011) |
Fosamax (alendronate Na 70 mg, PO weekly, 2014.01–2020.11) Prolia (denosumab 60 mg SC every 6 months, 2021.06–2024.01) |
Persistent pain and swelling (2015.03) |
Explantation of #37 (2015.11) Explantation of #36, extraction of #35, and sequestrectomy, Mn. Lt. (2016.03) Sequestrectomy, Mn. Lt. (2016.11) |
Complete mucosal healing after sequestrectomy |
| 2 | #44–#46 (2017) |
Bonviva (ibandronate 150 mg, PO monthly, 2018.11–2020.12) Evenity (romosozumab SC monthly, 2021.05–2022.08) Prolia (denosumab 60 mg SC single dose, 2022.09) |
Pricking pain and discomfort of the right lower lip and mandible (2021.01)** |
Sequestrectomy with fixture removal, Mn, Rt. (2021.02) Sequestrectomy, Mn, Rt. (2021.06) |
Complete mucosal healing after sequestrectomy (twice) |
| #25 (unknown) |
Stabbing pain and pus discharge (2023.09) |
Fixture removal (#25) (2023.12) | Pain persisted with pus discharge | ||
| #26 (unknown) |
Severe mobility and pus discharge (2024.04) |
Fixture removal (#26) (2024.04) | Pain persisted with pus discharge | ||
| #24, #36 (unknown) |
Stabbing pain and severe mobility (2024.07) |
Explantation of #24, #36 and sequestrectomy, Mx. & Mn. Lt. (2024.08) |
Complete mucosal healing after sequestrectomy |
Ⅲ. Discussion
Peri-implantitis is an inflammatory condition of the peri-implant mucosa that leads to progressive bone loss around dental implants.12 MRONJ is associated with antiresorptive or antiangiogenic therapy, with invasive dental procedures being major triggering factors. According to the 2022 AAOMS position paper, the incidence of MRONJ following implant placement remains undetermined.11 However, recent studies, including the Italian position paper, have reported that delayed or implant-associated MRONJ can occur even in patients receiving low-dose bisphosphonates, although such events are rare.10
Jung et al. observed similar implant success rates between patients with and without bisphosphonate exposure (97.3% vs. 98.2%).2 Likewise, the incidence of implant-related MRONJ in patients treated with denosumab for osteoporosis has been reported to be negligible.13 However, because bisphosphonates can remain attached to bone for extended periods and many patients receiving denosumab may have a history of bisphosphonate therapy, multiple factors may contribute to MRONJ development. Therefore, clinicians should provide patients with comprehensive counseling regarding potential risks, considering treatment duration and cumulative drug exposure.
Song et al. reported that bone resorption may be suppressed even in the presence of inflammation at the apex when zoledronate is administered,14 as bone resorption is inhibited by the action of bisphosphonates. In Case 1, mild osteosclerosis was observed around the implant, and pain persisted despite continuous treatment. Implant placement was performed during antiresorptive therapy; therefore, we cautiously speculated that a Stage 0 lesion may have been present around implant #37 and subsequently progressed following implant removal. Alternatively, in the case of implant #37, pain may have occurred due to peri-implantitis, with MRONJ developing secondarily after implant removal. In such situations, clinicians should recognize that implant removal, similar to tooth extraction, can cause MRONJ or worsen the lesion, and patients should be informed of this risk before the procedure.
In Case 2, the patient was not receiving any osteoporosis treatment at the time of implant placement. The prosthesis was removed after 1 year of osteoporosis therapy because of discomfort. Therefore, a direct association between antiresorptive medication use and implant failure cannot be established. However, when prescribing osteoporosis medications, clinicians should consider and discuss with patients the potential risk of implant-triggered MRONJ.
According to Jung et al.,2 the pathophysiology of implant-related sequestrum differs from that of peri-implantitis. In typical peri-implantitis, endotoxins secreted by pathogens activate an immune response that increases osteoclastic activity through various inflammatory cytokines while suppressing osteoblastic activity, ultimately leading to bone resorption. In contrast, when bisphosphonates are administered, osteoclastic activity and angiogenesis decrease due to the accumulation and subsequent release of bisphosphonates from the bone. Impaired wound healing and bone remodeling, caused by soft tissue toxicity and ischemic changes, can then result in necrosis of the surrounding bone, including the implant fixture, rather than the previously osseointegrated area.
The relationship between antiresorptive therapy and implant-related MRONJ remains controversial; however, emerging evidence suggests that a connection cannot be entirely ruled out.5, 9 Careful patient selection, early diagnosis, and appropriate surgical management, including reconstruction when indicated, are essential to minimize risk. Further studies are needed to establish evidence-based guidelines for implant placement and maintenance in high-risk patients.