Ⅰ. Introduction
Dental implants have become a well-established and predictable1 treatment modality for replacing missing teeth. As clinical confidence in implant success has grown, indications have expanded to include immediate implant placement and ridge preservation in sites compromised by endodontic and periodontal pathology.2,3 The prevalence of implant therapy in the United States increased at an average annual rate of 14% from 1999–2000 to 2015–2016,4 with countries such as the Republic of Korea exhibiting among the highest global penetration rates.
Despite high survival rates, dental implants remain susceptible to biological and mechanical complications. Among these, peri-implantitis is particularly concerning due to its accelerative and unpredictable progression.5 Its prevalence has been reported at 9.25% per implant and 29.48% per patient.6 This may relate to the weaker soft tissue seal around implants, where collagen fibers run parallel to the surface,7 rather than perpendicular as in natural teeth, reducing the barrier function. Additionally, while a rough surface enhances osseointegration, it complicates mechanical debridement in peri-implantitis cases. If left untreated, peri-implantitis can lead to implant loss, complex and time-consuming retreatment, and diminished patient quality of life.
Retrograde peri-implantitis (RPI) is a less common variant of peri-implant disease, characterized by a radiolucent lesion at the implant apex with intact crestal bone. Clinically, RPI may resemble periapical pathology and typically manifests during the early phase of osseointegration or shortly after prosthetic loading. First described by McAllister et al. in 1992,8 RPI has a reported prevalence ranging from 0.26%9 to 7.8%.10 Although relatively rare, its multifactorial etiology warrants attention.9
This report presents a clinical case of RPI in the mandibular canine region, successfully managed using a reconstructive surgical approach.
Ⅱ. Case Report
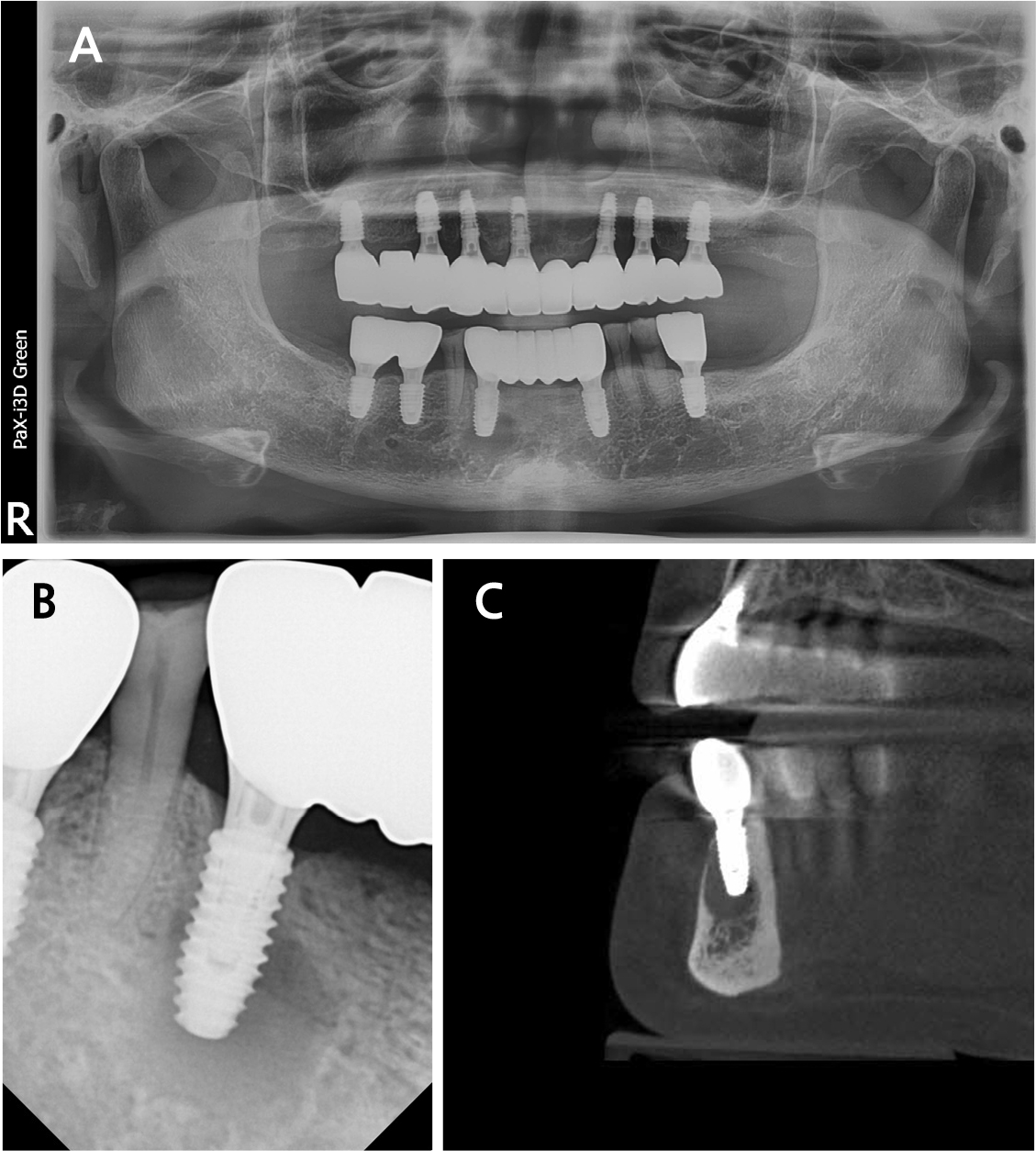
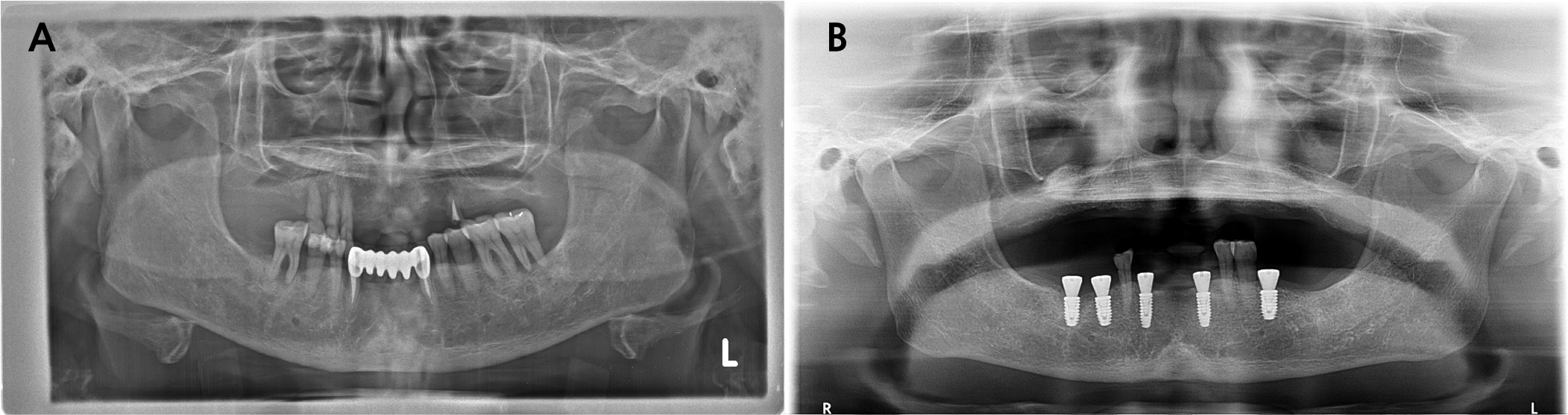
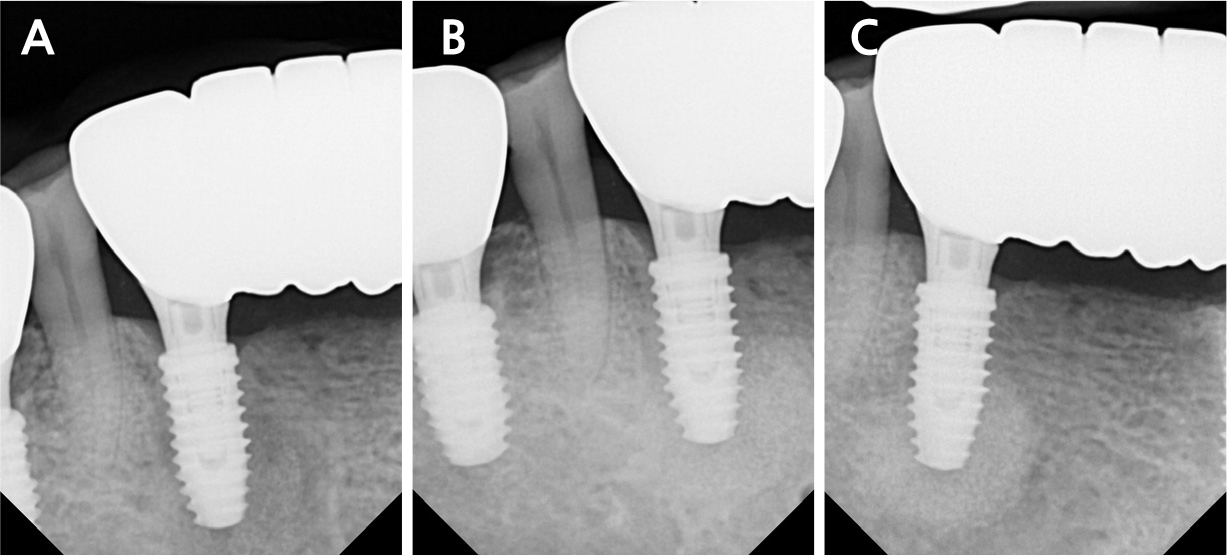
A 62-year-old female presented to the Department of Periodontology in Dankook University Dental Hospital with complaints of tenderness and a fever-like sensation around implant #43. A sinus tract was noted at the mesial aspect of the buccal peri-implant mucosa. Pocket probing depth ranged from 1–3 mm without bleeding. Panoramic and intraoral radiographs, along with cone-beam computed tomography (CBCT), showed intact crestal bone, a periapical radiolucency mainly involving the buccal aspect and extending lingually, and a thinned buccal bone with a 2 mm fenestration (Fig. 1). Adjacent tooth #44 responded normally to cold and electric pulp tests. The patient had a history of rheumatoid arthritis and allergies to sulfonamides and Penicillin. Her dental history included root canal treatment on #43, extractions owing to advanced bone loss (#15–13, 24, 33, 36, 37, 43, 45, 46), and implant treatment performed 1.3 years earlier using static computer-assisted surgery (Neo NaviGuide; Neobiotech, Wonju, Korea) (Fig. 2). A provisional diagnosis of RPI was made. A reconstructive surgical approach via the buccal aspect was planned.
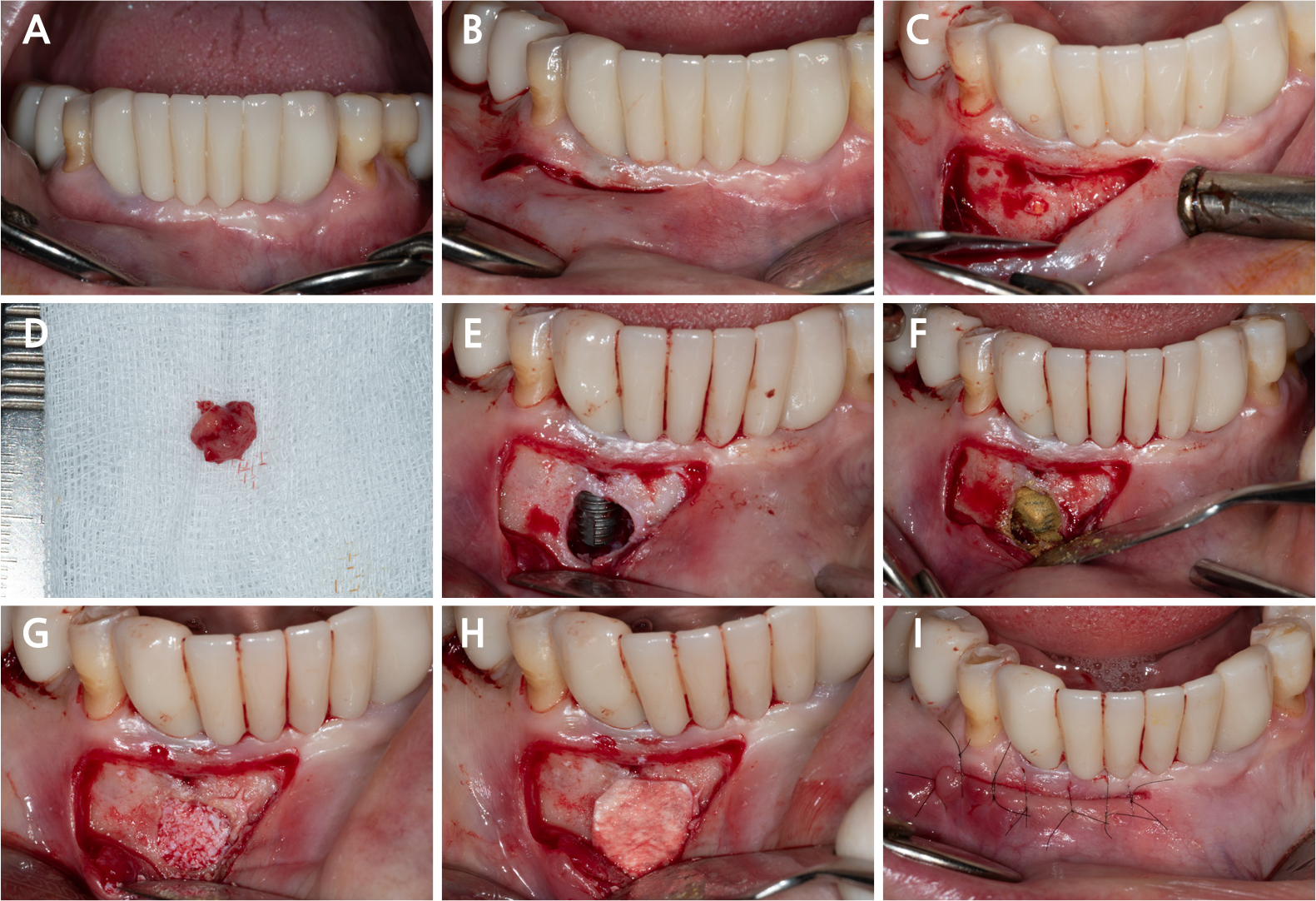
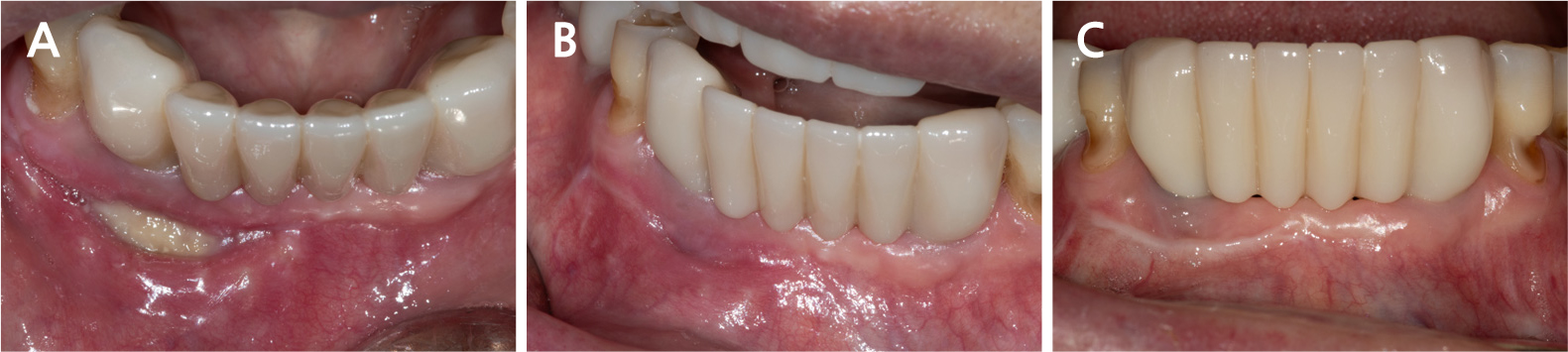
All surgical procedures were performed under local infiltration anesthesia using 2% lidocaine with 1:80,000 epinephrine. Fig. 3A shows the preoperative view. A horizontal incision with a vertical releasing incision at the distal end was made, including one or two adjacent tooth/implant crowns at the mucogingival junction (MGJ), using a #15c blade (Fig. 3B). Following mucoperiosteal flap elevation, the buccal fenestration (Fig. 3C) was apically expanded to approximately ∅5 mm circular window using a diamond drill from a sinus floor elevation kit (DASK Simple kit; Dentium, Suwon, Korea). Granulation tissue was detached and debrided using a surgical curette and hand instruments from the DASK Simple kit (Fig. 3D). The implant surface was decontaminated with a titanium brush (TN brush; Dentium) at 800 rpm under saline irrigation, followed by scrubbing with a cotton pellet soaked in tetracycline hydrochloride for 2 min (Fig. 3E and 3F). The peri-implant bone defect was filled with deproteinized porcine bone mineral (Osteon Xeno; Dentium) and covered with a porcine collagen membrane (Collagen Membrane II; Dentium) (Fig. 3G and 3H). Interrupted sutures were placed using a 6–0 monofilament nylon thread (Ethilon; Ethicon, Somerville, United States) to stabilize the wound (Fig. 3I). The patient was advised to avoid mechanical plaque control at the surgical site and to rinse twice daily with an antiseptic mouthwash (GUM dental rinse; Sunstar, Etoy, Switzerland) for 2 weeks. Antibiotics (clindamycin 150 mg, three tablets daily) and an analgesic (acetaminophen 600 mg, three tablets daily) were prescribed for 5 days to manage postoperative infection and pain. Symptoms resolved following surgery. The wound showed secondary healing at suture removal (2 weeks after surgery) and was fully closed by 4 weeks (Fig. 4). Radiographic bone fill was maintained at the 1.5-year follow-up (Fig. 5).
Ⅲ. Discussion
According to the 2017 World Workshop, peri-implantitis involves mucosal inflammation and progressive bone loss.10 RPI is a distinct form, characterized by apical bone loss with intact crestal bone and supracrestal soft tissue, and requires surgical access for decontamination.
RPI-associated bone defects often present with contained and favorable morphologies for reconstructive treatment. However, surgical accessibility depends on factors such as buccal bone thickness, implant location, and anatomical structures (e.g., mental foramen, mandibular canal). In this case, the lesion was located in the canine region with a buccal fenestration, allowing access through a buccal window.
Chan et al. reviewed various etiologic factors proposed for RPI,9 which can be categorized into four groups:
1. Infection-related factors: residual bacteria at the implant site, adjacent endodontic lesions, residual root particles or foreign bodies, and drainage of inflammation via marrow spaces.
2. Surgery-related factors: violation of the minimal distance from adjacent teeth, implant surface contamination, surgical drilling beyond implant length, vestibular bone fenestration, bone compaction, bone loss due to mucoperiosteal flap elevation, and premature loading.
3. Patient-related factors: poor bone quality and development of osteomyelitis.
4. Material-related factor: surgical technique specific to the implant system used.
In this case, residual bacteria at the implant site and the surgical technique associated with the implant system were suspected contributing factors. A retrospective study reported that placing implants in sites with residual endodontic pathology significantly increases the risk of RPI (odds ratio = 7.2).11 The implant at site #43 was placed in a healed ridge following the extraction of a tooth previously treated endodontically. Subsequently, an enlarging apical radiolucency was detected without clinical symptoms, highlighting the importance of regular radiographic monitoring. Incomplete lesion removal at the time of extraction may have contributed to its persistence. Furthermore, the use of static guided surgery may limit irrigation and elevate osteotomy temperature. An in vitro study found that static surgical guides resulted in an approximately 1°C higher temperature than open surgery, with temperature significantly affected by osteotomy depth and irrigation fluid temperature, especially in high-density bone.12 Therefore, strategies to enhance cooling during osteotomy should be considered to reduce RPI risk. These may include integrating irrigation ports into the surgical guide. The flap design in this case included an MGJ incision, which may have contributed to secondary healing. In periodontal regenerative techniques without papilla incisions,13 incisions are made apical to the MGJ in the mucosa, and horizontal mattress and interrupted sutures are used for primary closure. In this case, tissue characteristics resulting from the MGJ incision may have hindered proper flap eversion and tension-free closure.
Implant surface decontamination was performed using a titanium brush and tetracycline-soaked cotton pellets. The titanium brush has been reported effective in cleaning rough implant surfaces.14However, owing to limited access to the lingual aspect of the implant, tetracycline was additionally used. Tetracycline provides antimicrobial activity and minimal impact on SLA surface roughness,15though high concentrations may alter the titanium oxide layer16 and potentially affect re-osseointegration.17Romeo et al.,18 used metronidazole gel followed by tetracycline hydrochloride in resective surgery for the treatment of peri-implantitis. They found that adding implantoplasty to this protocol resulted in better clinical outcomes compared to resective surgery with chemical decontamination alone, highlighting the importance of mechanical surface modification.
Despite clinical improvement, this case highlights several limitations: (1) mechanical decontamination of the lingual surface was incomplete; (2) secondary wound healing occurred owing to the incision design; (3) histologic confirmation of regeneration was not performed; and (4) as a single-case report with short-term follow-up, the findings are inherently limited in generalizability. Although RPI is relatively uncommon, further well-designed prospective studies with larger sample sizes are needed to validate these findings.
Ⅳ. Conclusion
RPI can be effectively managed with a reconstructive surgical approach, provided that preoperative imaging confirms favorable defect morphology and accessibility. When considering a reconstructive approach for RPI, preoperative CBCT evaluation is essential to determine whether access to the implant surface is feasible, based on surrounding soft and hard tissues and anatomical structures. To reduce RPI risk, clinicians should ensure complete removal of endodontic lesions at the time of extraction and employ gentle osteotomy with sufficient irrigation to prevent thermal injury.