Ⅰ. Introduction
Ⅱ. Case Report
1. Informed consent was obtained from the patient for this case report
2. Implant placement and lateral bone augmentation
3. Postoperative healing, uncovering procedure, and prosthetic delivery
4. Surgical re-entry
5. Histopathological finding
6. Follow-up
Ⅲ. Discussion
Ⅳ. Conclusion
Ⅰ. Introduction
Peri-implant inflammation typically progresses in an orthograde direction, extending from the coronal to the apical region.1,2 Depending on the affected tissue, this condition can be classified as peri-implant mucositis or peri-implantitis. These conditions are primarily triggered by a bacterial infiltration through the implant-mucosa interface. Extensive research has explored the etiology, epidemiology, and treatment of orthograde peri-implant inflammation.1,2,3,4,5,6,7
However, the progression of peri-implant inflammation is not necessarily orthograde; several studies have documented retrograde peri-implantitis.8,9 This type of inflammation closely resembles the periapical infection observed in natural teeth. Various factors can contribute to retrograde peri-implantitis, including overheating during osteotomy preparation, excessive compression, preexisting pathological conditions at the implant site, and placement of the implant near existing infections.10 Among these, endodontic pathology (either at the implant site or in adjacent teeth) has been identified as a significant contributing factor.10,11
Recent findings have demonstrated that peri-implant inflammation can also originate along the lateral surface of the implant, a condition referred to as lateral peri-implantitis.12 Current data on lateral peri-implantitis remain limited, and its etiology may differ from that of conventional peri-implantitis. A previous case report highlighted instances of lateral peri-implantitis in a posterior mandibular implant that had undergone bone augmentation. Inflammation developed 26 months after the implant placement. Surgical re-entry revealed extensive destruction of the previously augmented area. The authors hypothesized that the inflammation originated from the contamination of the graft material; however, the relatively delayed onset warrants further investigation.
Herein, we present a case of late-onset lateral peri-implantitis. Considering a lack of well-documented information on this condition, this case report provides valuable scientific evidence.
Ⅱ. Case Report
1. Informed consent was obtained from the patient for this case report
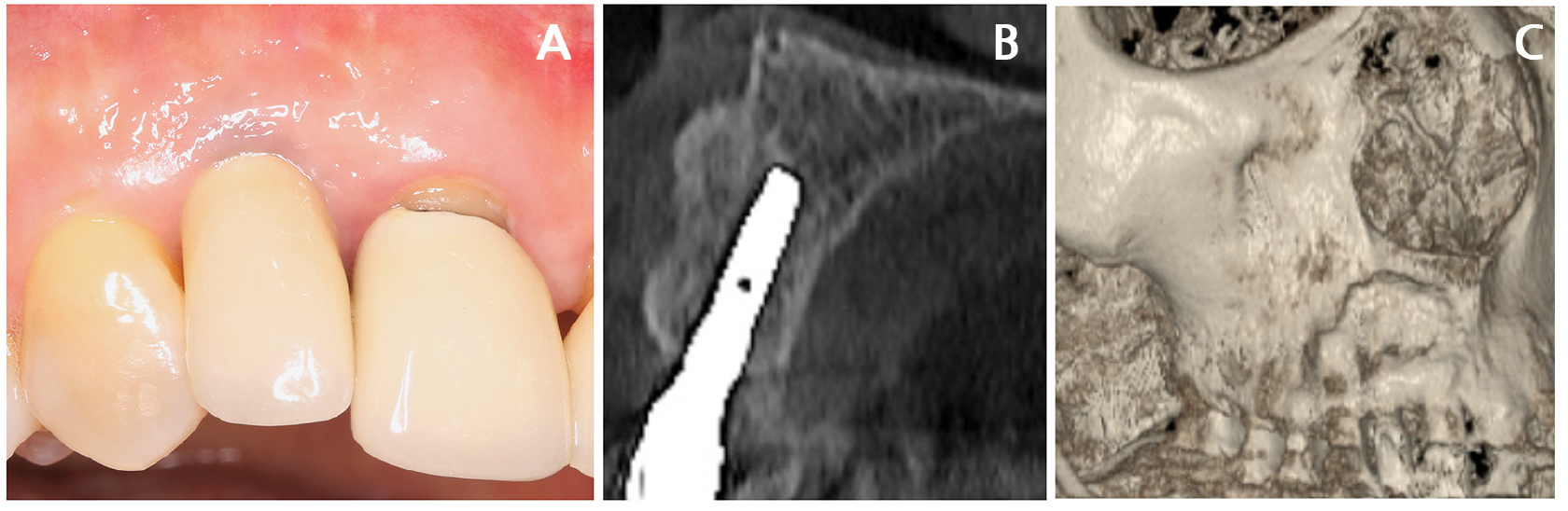
A 42-year-old non-smoking female visited a private clinic for implant treatment of a missing right maxillary lateral incisor. Panoramic and cone-beam computed tomography (CBCT) revealed an atrophic bone plate at the #12 site (Fig. 1A and 1B). The tooth was extracted 1 year prior. Lateral bone augmentation was scheduled simultaneously with the implant placement.
2. Implant placement and lateral bone augmentation
Under local anesthesia with lidocaine containing 1:100,000 epinephrine (Huons, Seoul, Korea), a mid-crestal incision and two vertical incisions were made, followed by full-thickness flap elevation. Labial bone depression was observed. Following osteotomy according to the manufacturer's guidelines, intramarrow penetration was performed using a low-speed #6 round bur to stimulate the osteogenic source. The depth of penetration was approximately 2–3 mm. Subsequently, a sandblasted, large-grit, acid-etched-textured implant was placed (Ø 3.8 × 12 mm, Implantium; Dentium, Suwon, Korea), followed by the connection of a cover screw (Fig. 1C, 1D, and 1E). A synthetic bone-substitute material (Osteon III; Genoss, Suwon, Korea) was grafted onto the labial side to improve the tissue contour (Fig. 1F). The bone-substitute material consisted of 60% hydroxyapatite and 40% beta-tricalcium phosphate. Subsequently, a crosslinked collagen membrane (Genoss) was used to fully cover the grafted area (Fig. 1G). After periosteal releasing incision, primary flap closure was achieved using a 4-0 nylon suture (Ethilon® 4.0; Ethicon, Cincinnati, United States) (Fig. 1H). A systemic antibiotic (Cefradine 500 mg; Yuhan Pharmaceutical Co.; Seoul, Korea) and a nonsteroidal anti-inflammatory drug (Etodol® 200 mg; Yuhan Pharmaceutical Co.) were administered thrice daily for 10 days. The patients were instructed to rinse their mouths with 0.12% chlorhexidine solution (Hexamedine; Bukwang Pharmaceutical, Seoul, Korea) for 2 weeks. The sutures were removed 10 days after surgery.
3. Postoperative healing, uncovering procedure, and prosthetic delivery
No adverse events were observed during the healing period (Fig. 1I). Moreover, the patient reported no abnormalities. The implant site was uncovered after 5 months (Fig. 1J). The remaining collagen membrane was then removed from the augmented area. Probing revealed a soft consistency in the regenerated tissue, which was, however, well-integrated with the surrounding area. A healing abutment was connected to the implant (Fig. 1K). The flap was sutured using 4-0 catgut. After 2 months, the final prosthesis was inserted. A well-established bony contour was observed on the CBCT scan obtained immediately following the final prosthesis delivery (Fig. 2).

Fig. 1
Implant placement, lateral bone augmentation, and uncovering. (A) Panoramic radiograph before implant placement, (B) Labial bone deficiency observed in the coronal image of the cone-beam computed tomography scan, (C) clinical situation before implant placement, (D) after implant placement, (E) intramarrow penetration performed on the labial bone using a surgical round bur, (F) synthetic bone substitute material grafting, (G) A cross-linked collagen membrane is applied, and (H) primary flap closure is achieved, (I) Clinical situation after 5 months (at the time of uncovering), (J) Well-established tissue contour is observed, (K) 2 months after uncovering.
The patient was regularly followed up. On clinical examination at 4 years of age, healthy peri-implant tissue was observed. CBCT examinations at that time exhibited well-consolidated augmented bone, with relatively decreased radiopacity at the mid-portion of the augmented area compared to the coronal and apical surfaces. In the reconstructed three-dimensional image, a small irregular depression was observed at the apical region of the augmentation (Fig. 2). However, no further abnormalities were observed.
A 7-years post-implantation, the patient reported slight tenderness in the labial mucosa at the implant site. Mucosal recession (approximately 1 mm) developed in the labial area, and the metal collar was slightly exposed. The probing depth was less than 3 mm, with bleeding on probing noted in the midfacial area. On CBCT examination, depression of the facial contour of the augmented bone and further loss of radiopacity were observed. The reconstructed three-dimensional image demonstrated progression of the previously noted depression (observed at 4-years of follow-up). Compared to the clinical photographs taken at the time of surgery revealed that the site of depression corresponded to one of the intramarrow penetration holes (Fig. 2).
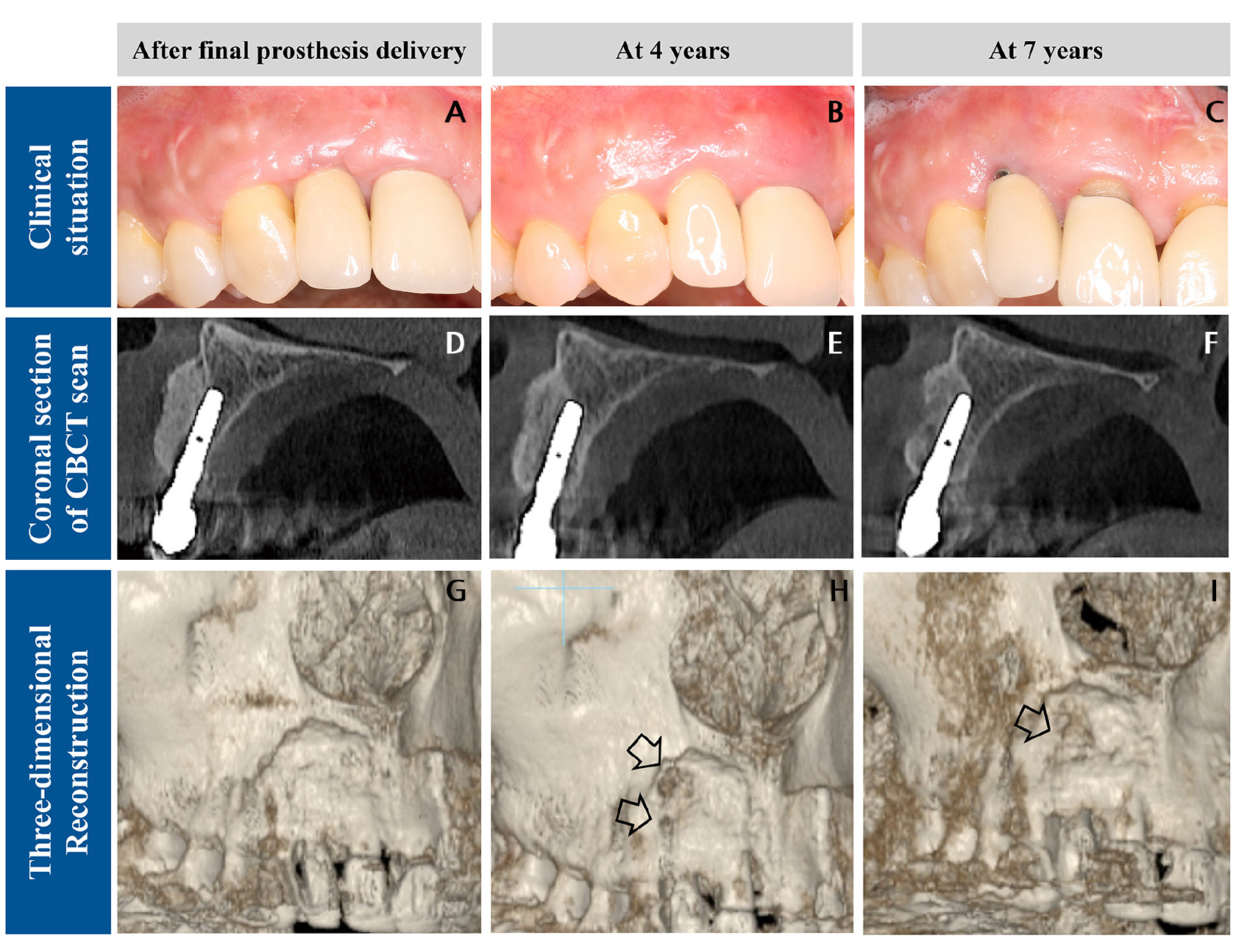
Fig. 2
Clinical and radiographic characteristics during recall visits. (A, B, C) Clinical conditions, (A) immediately following final prosthesis delivery, (B) at 4 years, (C) at 7 years, (D, E, F) coronal sections of the cone-beam computed tomography (CBCT) scans, (D) immediately after final prosthesis delivery, (E) at 4 years, (F) at 7 years. (G, H, I) Three-dimensional reconstruction of the CBCT images, (G) immediately after final prosthesis delivery, (H) at 4 years, and (I) at 7 years. The arrows indicate bone depression in the reconstructed images.
4. Surgical re-entry
Based on the pathological changes in the CBCT scans, the attending dentist (W.B.-P.) decided on a surgical intervention. After flap reflection, granulation tissue occupied approximately two-thirds of the implant surface and neighboring areas (Fig. 3A). The granulation tissue and disintegrated bone-substitute particles were thoroughly removed using a curette. Subsequently, some augmented tissues were also removed (Fig. 3B). The excised specimens were fixed in 10% formalin for histopathological examination. The coronal and palatal bone surrounding the implant body remained relatively intact (as determined by the tissue consistency during debridement). The implant did not demonstrate mobility or rotation after granulation tissue removal, supporting the decision to maintain it.
The exposed implant surface was debrided using titanium curettes and a titanium brush (TN-Brush, Dentium) to ensure thorough surface decontamination. The surface was then treated with cotton pellets soaked in 100 mg/mL tetracycline-hydrochloride (HCl) solution (Tc-HCl 500 mg powder mixed with 5 mL physiological saline solution) for 5 minutes (Fig. 3C). Finally, the implant surface and defects were meticulously irrigated with saline solution.
The defect was filled with a bovine bone-substitute material (OsteonXeno; Genoss) (Fig. 3D). However, barrier membranes were not applied. To prevent soft tissue shrinkage, a subepithelial connective tissue graft was harvested from the lateral palate and applied to the surgical site (Fig. 3E). The connective tissue graft was not anchored with sutures but was well-adapted. The flap was repositioned and secured using 4-0 black silk sutures (Fig. 3F). The same postoperative regimen was employed for implant placement and bone augmentation. Furthermore, healing was uneventful.
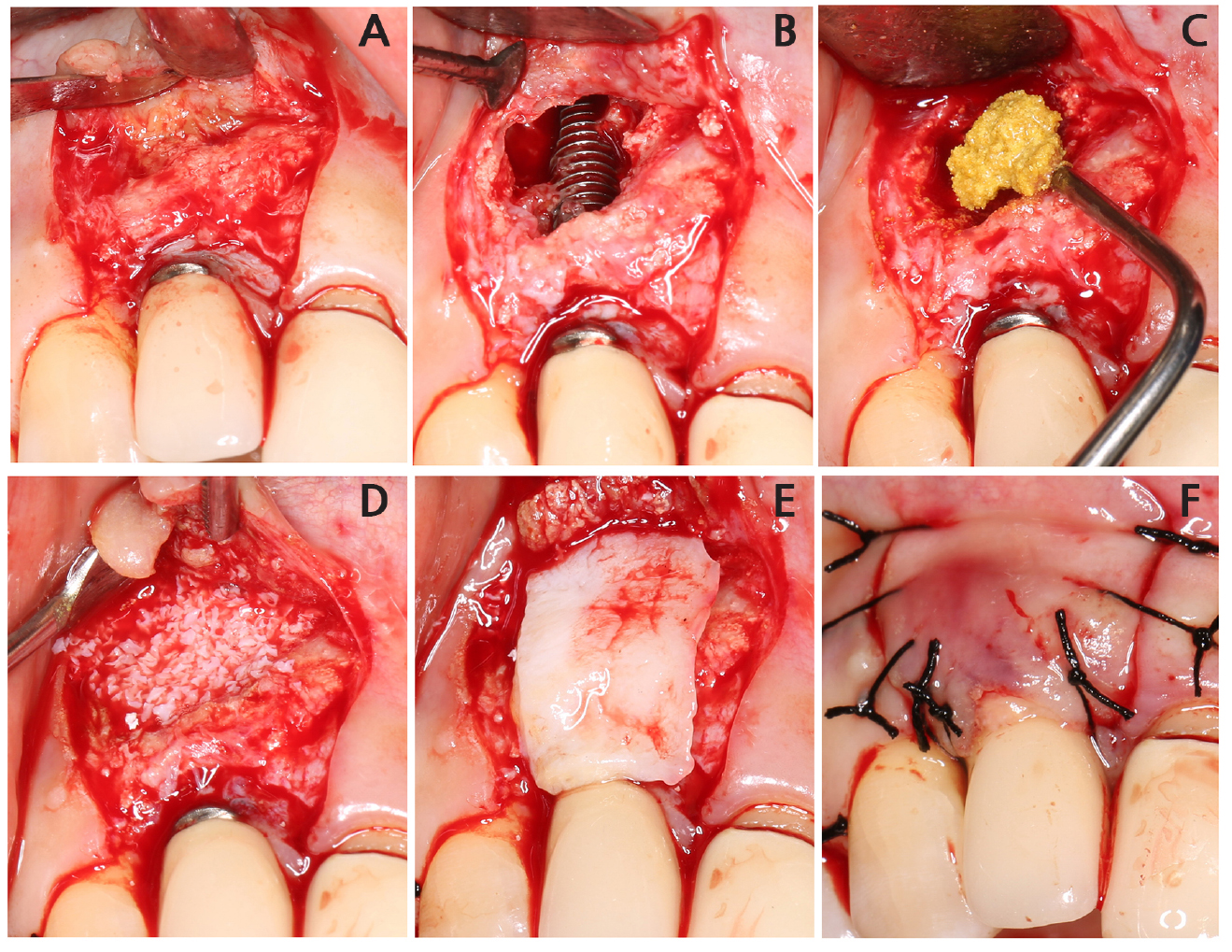
Fig. 3
Re-entry. (A) The labial flap is reflected, and (B) the implant body is exposed after removing the granulation tissue and disintegrated bone graft particles. The crestal and apical portions of the implant bodies are observed to be relatively intact. Mechanical debridement is performed on the implant surface using a titanium brush and titanium curette, (C) Tetracycline-hydrochloride is applied for chemical decontamination, (D) The bone defect is filled with xenogeneic bone substitute material, (E) A subepithelial connective tissue graft is applied, (F) The flap is closed.
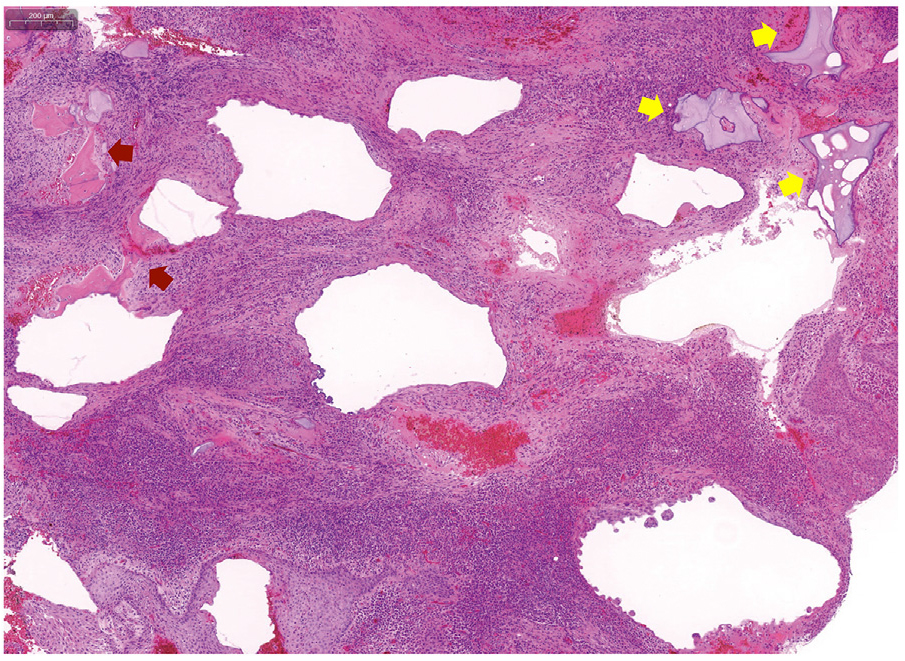
5. Histopathological finding
The retrieved specimens were stained with hematoxylin and eosin. The specimens revealed fibrotic tissue primarily with abundant inflammatory cell infiltration. Small fragments of vital (lacunae and osteocytes) and necrotic bone were observed (Fig. 4).
6. Follow-up
The patient was followed up twice a year after re-entry. At the 1-year follow-up examination, no signs or symptoms of inflammation were detected. Additionally, mucosal recession was also resolved (Fig. 5A). The CBCT scan demonstrated a reestablished bone contour (Fig. 5B and 5C). No specific inflammatory events were observed up to the two-year follow-up, and monitoring was continued thereafter.
Ⅲ. Discussion
Orthograde progression is a common feature of peri-implant inflammation. However, only a small number of patients exhibit progression in the opposite direction. Previously, the prevalence of retrograde peri-implantitis was reported to be 0.26–1.86%.13,14 This low prevalence may be attributable to a significant lack of awareness regarding retrograde peri-implantitis.15 In this study, we present a potentially unrecognized dimension of peri-implant inflammation: lateral peri-implantitis.
Until noticing lateral peri-implantitis, it took approximately 7 years for the present patient. This is a somewhat unique property of lateral peri-implantitis in this case study. Retrograde peri-implantitis is known to occur within 6 months of implant placement, as the condition is mainly related to the apical pathology of the adjacent tooth and residual inflammation at the implant site.16 Consistent with these findings, if an infection had occurred during implant placement or bone augmentation, clinical and radiographic signs or symptoms would likely have manifested during the early healing phase. However, based on CBCT scans, inflammatory signs were not evident during the early healing phase or up to 4 years postoperatively. To further elucidate this condition, another case report of lateral peri-implantitis may provide valuable insights.12 In that study, the authors commented that the consistency of newly formed tissue was very soft (easily perforated by probing), which might indicate the presence of sub-clinical infection. In the present study, the consistency of the regenerated tissue was also soft 5 months post-augmentation. Even though this stage might be too early to establish a new cortical plate, sub-clinical infection could be suspected in the present case.
The position of intramarrow penetration appeared to correspond to the location of the defect. Intramarrow penetration serves to stimulate osteogenic cells. Intramarrow penetration has been demonstrated to enhance bone fill and density in bone regenerative procedures.17 To the best of our knowledge, no reports have documented adverse effects associated with this method. However, penetration holes may serve as conduits for transmitting infections.
In this case, the contaminated implant surface was mechanically and chemically disinfected. As bone augmentation was performed at the defect site, decontamination of the implant surface was crucial for re-osseointegration,18 irrespective of the material used for bone augmentation. Moreover, decontamination of moderately rough implant surfaces may not be easy.19 Although no single disinfection method or combination thereof has been proven superior, several studies have demonstrated the effectiveness of titanium curettes, titanium brushes, and Tc-HCl.3,20
The reaugmented area was well consolidated on the CBCT scan 1 year after reentry. However, close monitoring of the patient is essential due to the uncertain etiology of lateral peri-implantitis and its late development.