Ⅰ. Introduction
Alveolar ridge preservation (ARP) after tooth extraction has been known to be effective,1 but there is a lack of evidence of ARP after implant removal. Peri-implant disease is divided into peri-implant mucositis and peri-implantitis according to the presence or absence of supporting bone involvement, of which peri-implantitis has various diagnostic criteria and treatment options despite the high implant survival rate.2
Misch et al.3 diagnosed implant failure as functional pain, mobility, and bone loss of more than half the implant length on radiographs. If the existing implant should be removed according to these diagnostic criteria, immediate re-implantation wider than the existing implant is considered the most ideal alternative, and it has the advantage of minimizing bone resorption and reducing the treatment period and the number of surgeries.4 However, few statics compare three-dimensional changes5 and it is challenging to obtain osseointegration.6 Without bone grafting, enormous soft and hard tissue augmentation surgery will be required in the future, which increases the difficulty of the surgery and the patient's discomfort. ARP after extraction has recently attracted attention as a technique that can prevent these problems early,7 and preserved more keratinized mucosa without primary closure.8 However, only a few cases have been reported regarding the success of implants at the failed implant site using the ARP. Thus, this case report aimed to present a case of reimplantation using the ARP with an open-healing approach.
Ⅱ. Case Report
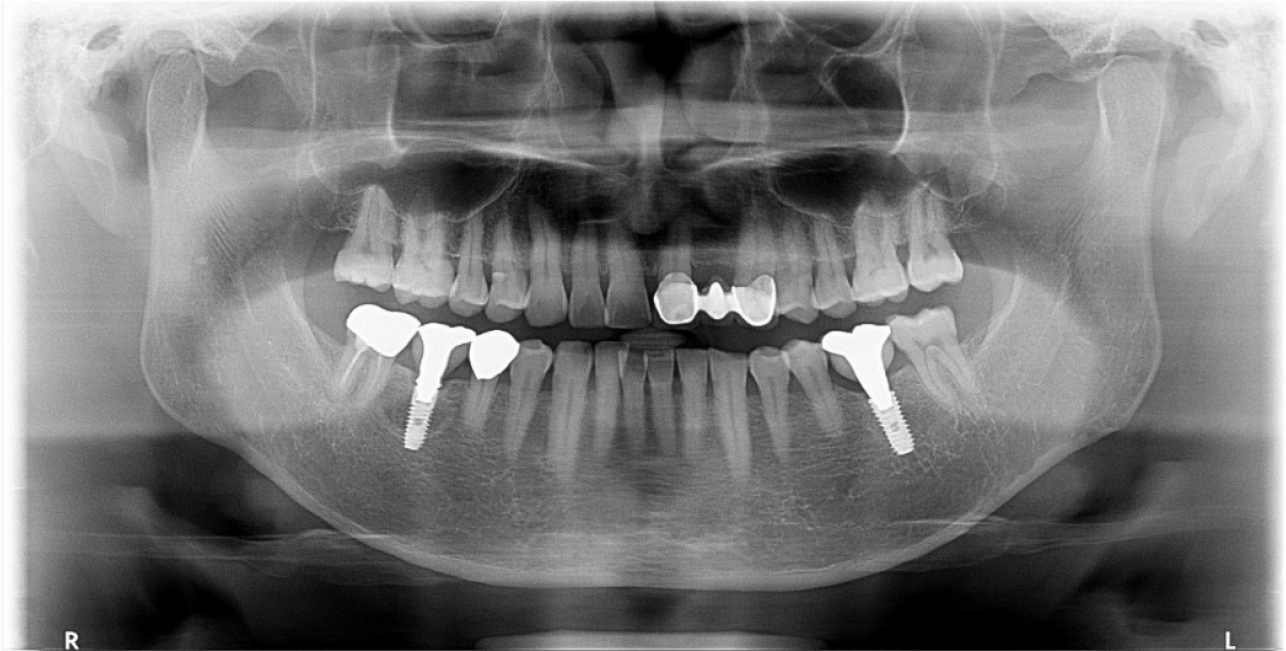
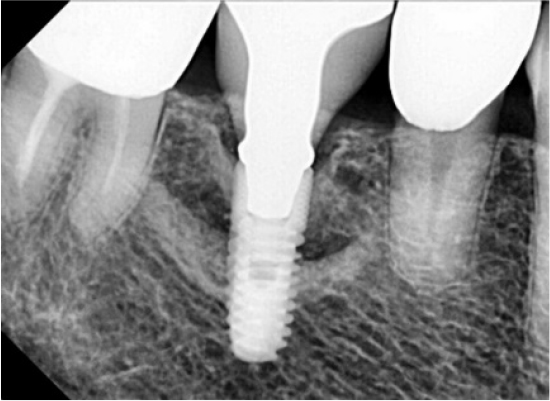
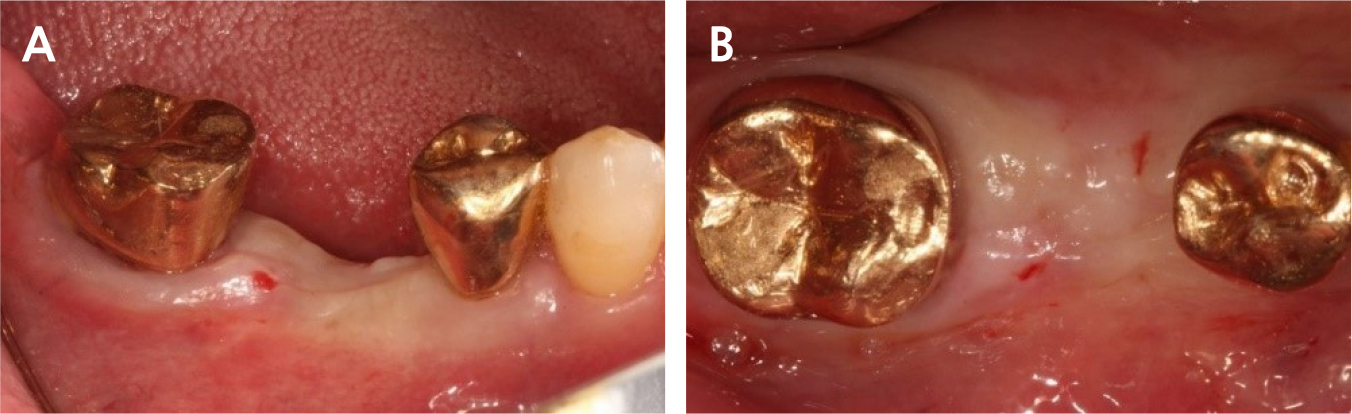
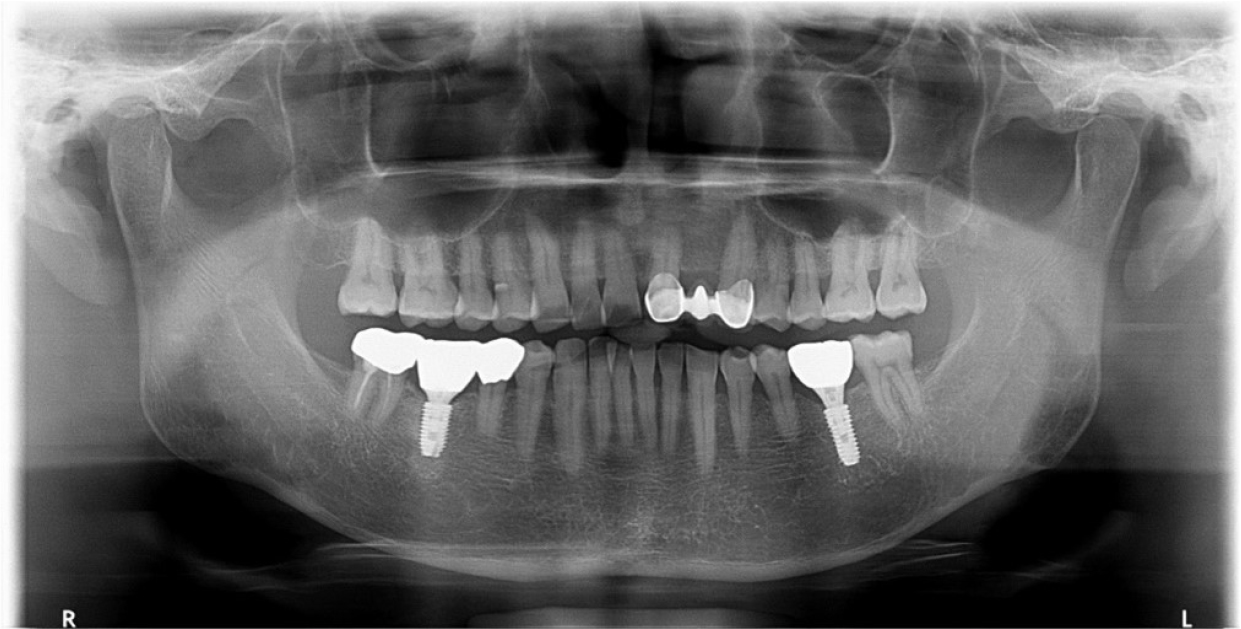
A 55-year-old man visited the clinic for implant discomfort and bleeding. The implant in the lower right 1st molar was placed three years ago at a private practice and was found to exhibit symptoms of buccal mucosal swelling and mesio-buccal deep probing pocket (Table 1). A sclerotic line on the fixture middle level was observed on panoramic and periapical radiographs (Figs. 1 and 2). There was no mobility or pus discharge but a throbbing pain during function. The probing depth was 10 mm; therefore, he was diagnosed with peri-implantitis, and we recommended implant removal. Patient hoped preservation, so regenerative therapy were preceded.
Table 1.
Patient’s information
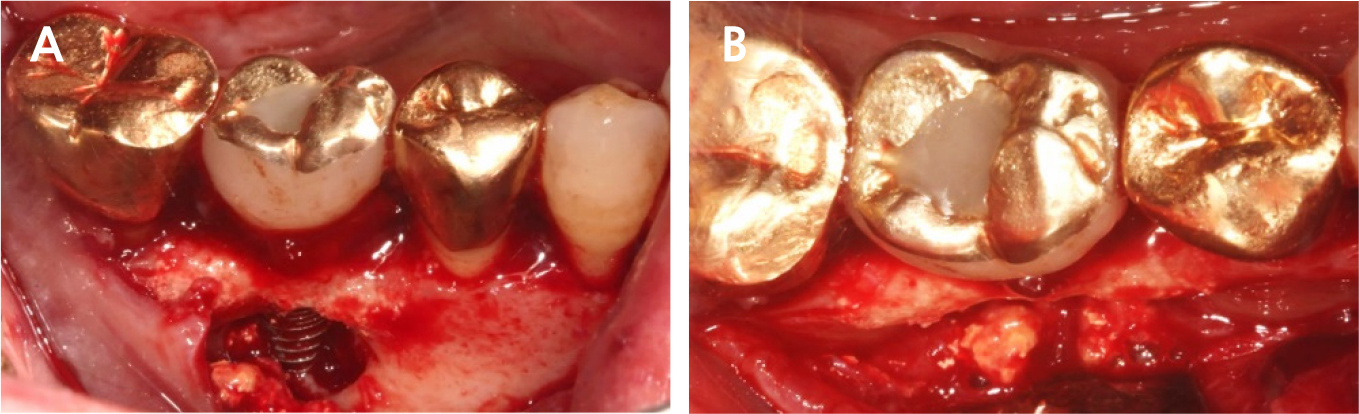
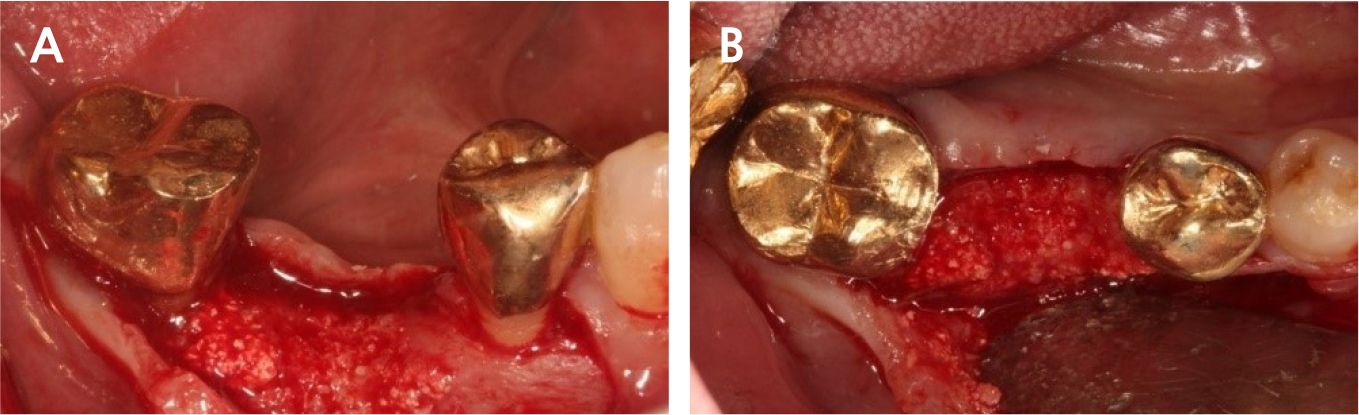
Under local anesthesia with 2% lidocaine containing 1:100,000 epinephrine (Huons, Seongnam, Korea), a buccal flap was elevated after a conventional circular incision. Most of the mid-buccal bone showed necrosis (Fig. 3). Decontamination was impossible due to the poor accessibility of the lingual part, including necrotic and inflamed portions; therefore, we decided to extract the implant using forceps.
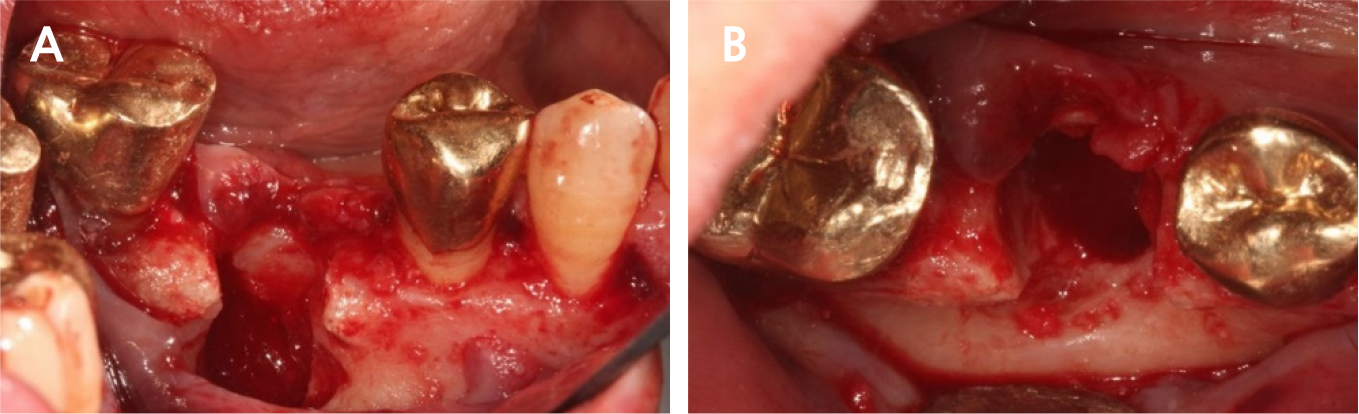
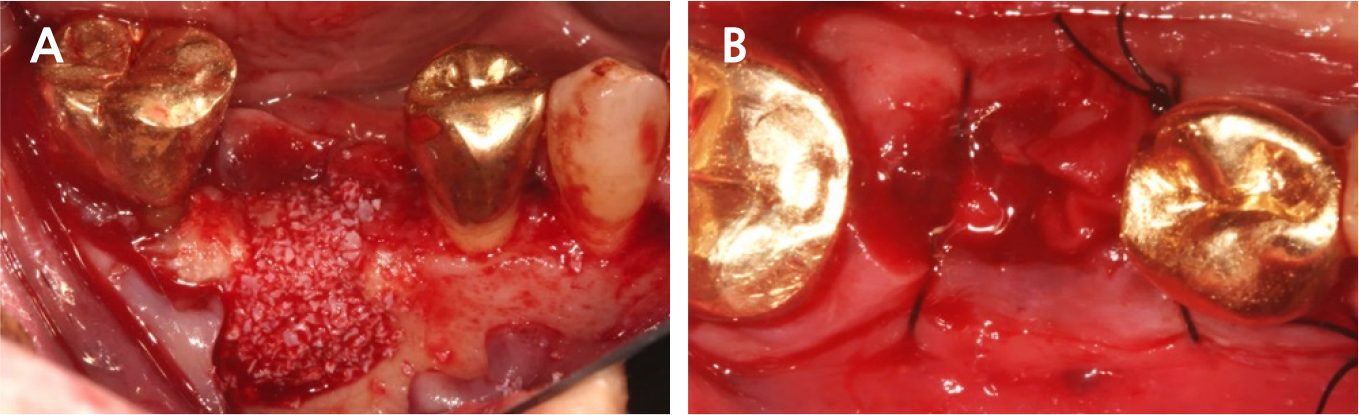
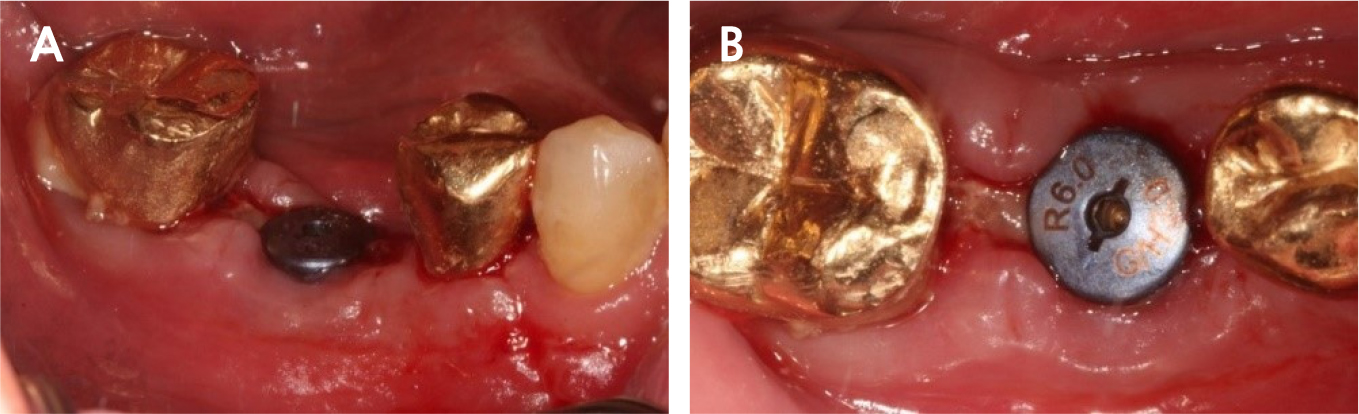
During implant removal, the buccal remnant marginal bone was brittle and easily fell off, and the large bone defect area needed additional treatment for delayed implant placement(Fig. 4). After augmentation with a combination of 0.25 g of deproteinized bovine bone mineral (DBBM) (Bio-Oss®; Geistlich, Wolhusen, Switzerland) and 250 mg of DBBM with 10% porcine collagen (Bio-Oss collagen®; Geistlich), double layer of native bilayer collagen membrane (Bio-Gide®; Geistlich, Switzerland) was applied. The socket was sutured in an open healing state using hidden X suture (Figs. 5 and 6).9
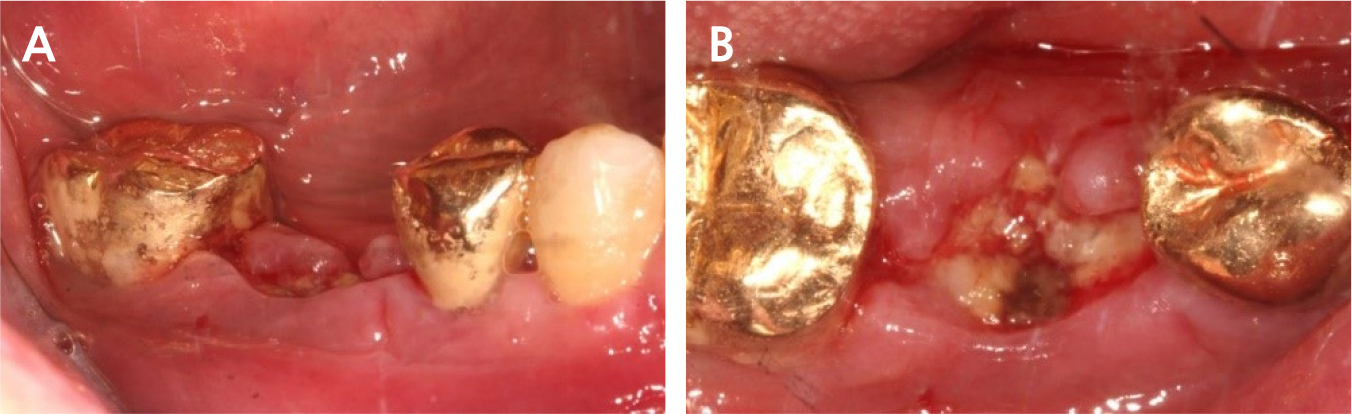
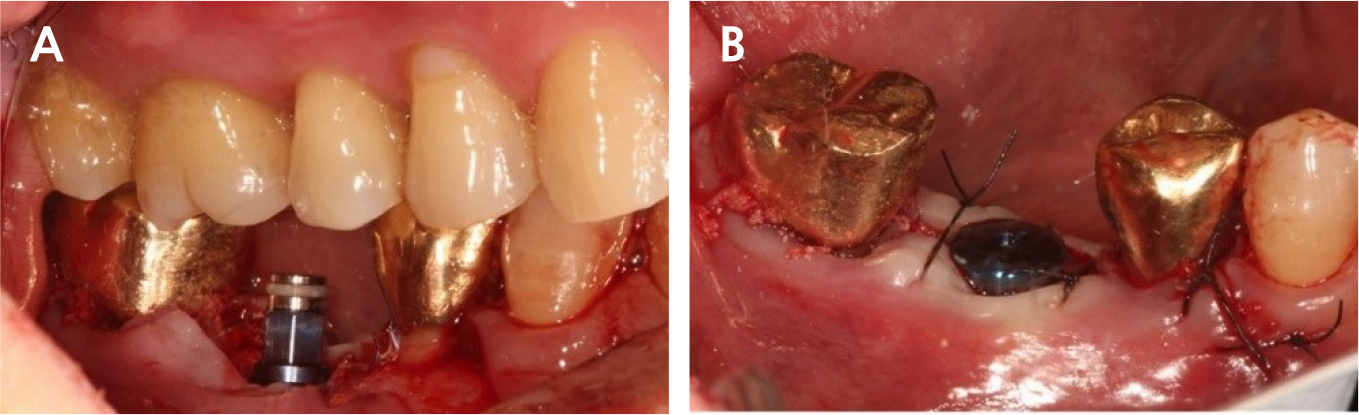
There was slightly sloughing tissue in the open healing area one month later, the healing of the socket seemed like a crater pattern. However, after 4 months, high-quality keratinized mucosa was secured (Figs. 7, 8, and 9). At the time of flap opening for implant placement, the bone was maintained at the ideal height and width. The bone quality was sufficient to get initial stability (Fig. 10).
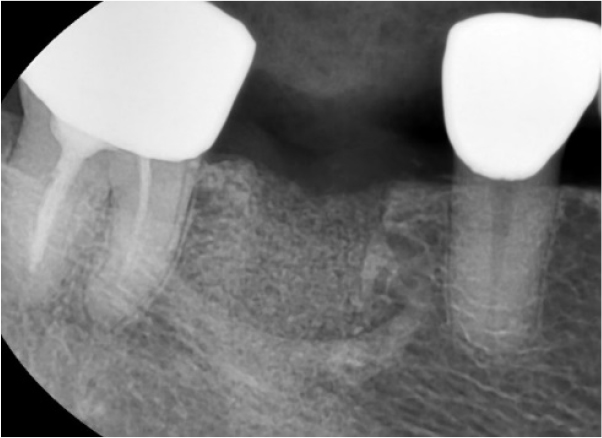
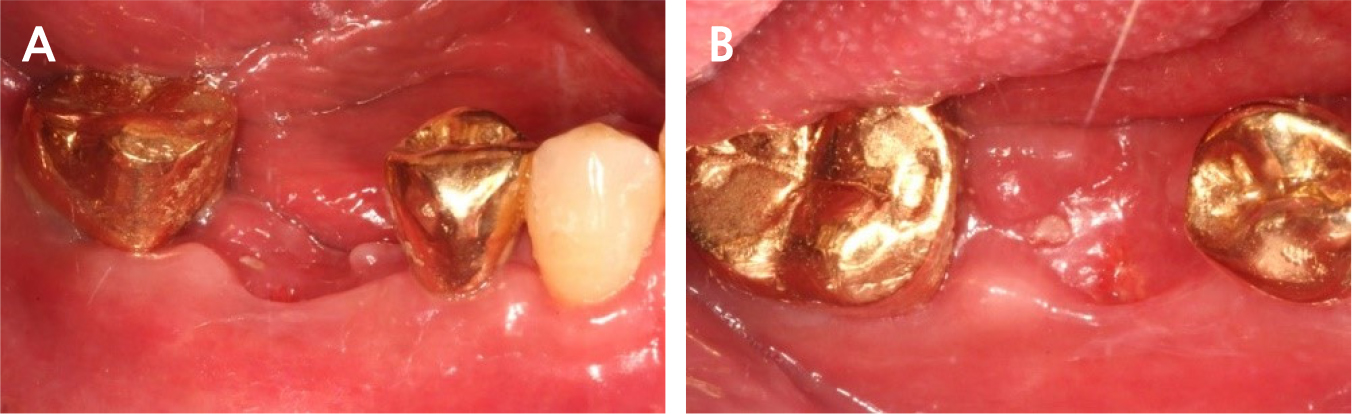
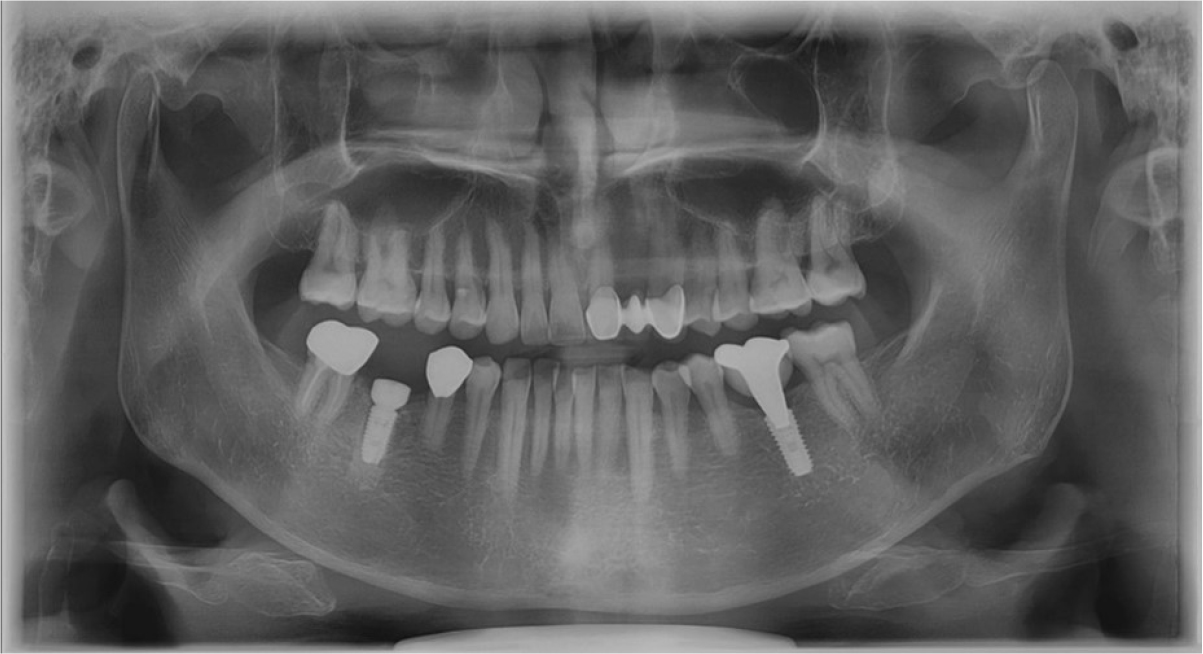
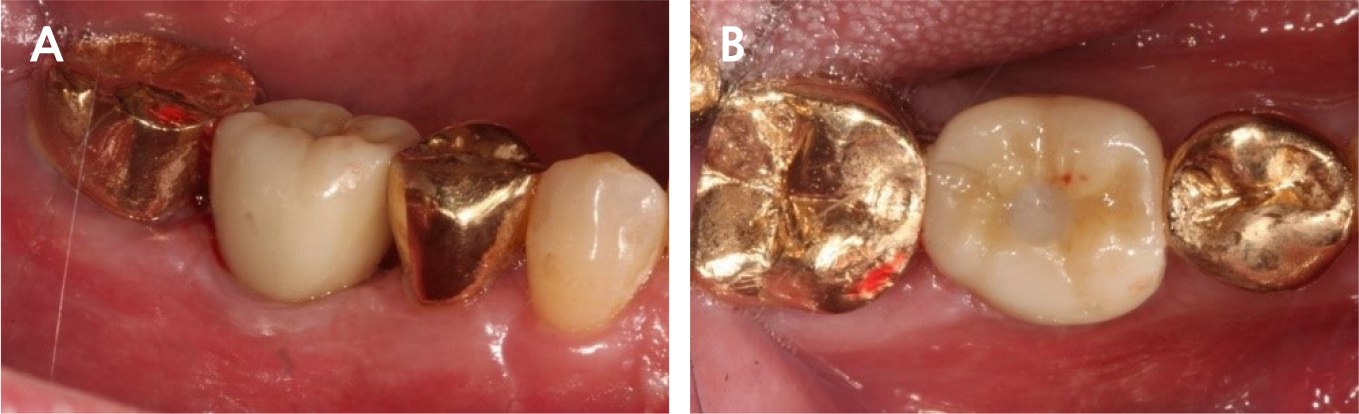
An implant of 5.0 mm diameter and 10.0 mm length (Luna®; Shinhung, Seoul, Korea) was placed at the lower right 1st molar site, and the implant stability quotient (ISQ) was 81. The Φ6.0 × 5.0 mm healing abutment was placed and sutured with 4-0 Supramid® (Assut Medical Sarl, Pully-Lausanne, Switzerland) (Fig. 11). It showed stability on the post-operative panoramic radiograph (Fig. 12). After 4 months, the final prosthesis was completed (Figs. 13 and 14).
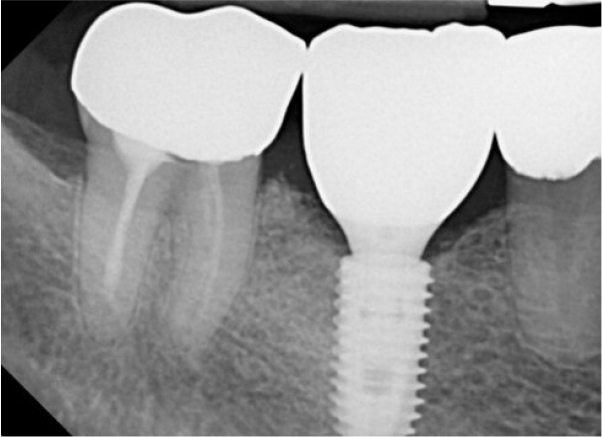
At 5 years follow-up, the implant showed stable alveolar bone without any signs of peri-implantitis or bone resorption on the radiograph analysis (Figs. 15 and 16). In addition, there was no bleeding on probing and no clinical problems.
Ⅲ. Discussion
Definite implant failure is defined as pain or mobility during use or radiological bone loss of >50% of the implant length. In this case, radiologic radiolucency revealed necrosis occurring in the lower part of the implant, such as apical lesions, It appeared as an implant apical lesion caused by remnant endodontic or apical lesions before previous implant placement10 or osseointegration failure of augmented bone.11 Mardinger et al.12 recommended removing a failing implant as soon as possible because it was advantageous for the success rate of reimplantation.
A second attempt at reimplantation at an implant failure site should be cautious because there is a lack of sufficient data on replacing failing implants.6 Bruno et al.5 reported significantly lower survival rates when reimplanted at the implant failure site, and Kim et al.13 reported lower survival rates when reimplanted for second, third, and fourth reimplantations at the failed site.
In cases of dehiscence or fenestration defects, guided bone regeneration can secure the stability of the graft material.14,15 Particulate bone showed more new bone formation than block bone.16 Block bone has advantages in terms of reduction of bone displacement and resistance to compression due to suture,17 but it has disadvantages in terms of reduction of horizontal thickness.18 To overcome this, a combination of particulate and block bone at guided bone regeneration stabilizes bone volume more.19
Grossmann et al.6 suggested that reimplantation at the same site should be performed within 9-12 months after the removal of a failed implant, and proper site conditions can increase the success rate of single dental implantation at the implant failure site. To reduce the failure rate, adequate keratinized mucosa and hard tissue should be needed.13 Schwarz et al.20 mainly classified peri-implantitis into two defect patterns: well-defined intra-bony defects (Class I defects) and horizontal bone loss (Class II defects). Implying that most of these defects require additional guided bone regeneration at the previously failed site.
Engler-Hamm et al.8 found that keratinized mucosa could be preserved more and that there was less discomfort and swelling after surgery using ARP without primary closure. When suturing an open-healing area, a bucco-lingual pulling vector occurs in the conventional X suture, but the Hidden X suture can maximize the amount of keratinized mucosa by pulling mesio-distally.9
This case presented a challenging situation to replant immediately after explantation due to poor residual buccal bone and insufficient keratinized mucosa; thus, an open-healing approach at the explantation site was performed to increase soft and hard tissue. Five-year follow-up of the lower right 1st molar, implants showed favorable conditions at the gingival margin and alveolar bone, with no signs of peri-implantitis.
Ⅳ. Conclusion
Implant installation at an implant failure site has a lower survival rate than that of the initial implant. ARP using open healing in an explantation site could be advantageous for replantation as an aspect of sufficient soft and hard tissue gain. Moreover , it can reduce inconvenience and difficulty of surgery for patients.