Ⅰ. Introduction
Ⅱ. Materials and Methods
1. Patients and ethical statements
2. Surgical procedure
3. Radiographic evaluation
4. Self-reported questionnaire and clinical examination
5. Statistical analysis
Ⅲ. Results
1. Baseline characteristics
2. Radiographic outcomes
3. Postoperative discomfort and wound healing outcomes
Ⅳ. Discussion
Ⅴ. Conclusion
Ⅰ. Introduction
A dental implant is one of the most useful treatment modalities for the replacement of missing teeth.1 A long-term retrospective study showed that the cumulative survival rates of implants in the lower posterior region and anterior teeth were 91.4% and 94.7%, respectively.2 Simonis et al.3 reported that the cumulative survival and success rates of long-term implants of 10 to 16 years was 82.94%. A recent systematic review and meta-analysis also confirmed that the 10-year survival rate at the implant level was 96.4% (95% confidence interval: 95.2% – 97.5%).4
Guided bone regeneration (GBR) is the most documented and commonly used approach for the augmentation of horizontal and vertical alveolar bone defects in the maxilla and mandible.5 Narrow diameter implants are frequently used in the mandibular anterior region because of anatomical constraints (such as limited buccal–oral dimensions), and GBR is often used for appropriate bone augmentation and adequate esthetic outcomes.6,7 In esthetically sensitive areas, GBR has the potential to reestablish the buccal and palatal/lingual contours of the alveolar ridge, thus helping the development of soft tissue around the implant.8
Although various bone grafting materials are currently used for GBR, data on the superiority of specific biomaterials are controversial. Moreover, achieving reliable and efficient bone augmentation remains challenging. Recently developed soft-type block bone substitutes are widely used in clinical practice, particularly in alveolar ridge preservation procedures.9,10 However, relatively few studies have focused on the clinical benefits of using soft-type block bone substitutes for peri-implant dehiscence defects. This study aimed to evaluate the clinical outcomes of porcine-derived, soft-type block bone substitutes in combination with a resorbable collagen membrane for the treatment of peri-implant dehiscence defects in the mandibular anterior region.
Ⅱ. Materials and Methods
1. Patients and ethical statements
The study protocol was approved by the Institutional Review Board of Daejeon Dental Hospital, Wonkwang University (approval no. W2105/001-001). The requirement for informed consent of all subjects was waived because of the retrospective nature of this study. This study enrolled 12 patients who underwent implant placement with GBR in the mandibular anterior region between September 2016 and May 2021 in the Department of Periodontology, Daejeon Dental Hospital, Wonkwang University.
2. Surgical procedure
A periodontist (JHL) performed all implant surgeries. Antibiotic (netilmicin 50 mg/2 mL) and analgesic (diclofenac 90 mg/2 mL) injections were administered to all subjects 30 min before implant surgery, and a course of antibiotic (amoxicillin 500 mg tid for five days) and analgesic (ibuprofen 200mg tid for five days) therapy and antimicrobial mouthwash rinse (0.12% chlorhexidine, bid for two weeks; Hexamedine, Bukwang, Seoul, Korea) were prescribed after the implant surgery. A local anesthetic (2% lidocaine HCl with 1:100000 epinephrine; Yuhan, Seoul, Korea) was delivered at each surgical site. A mid-crestal incision with or without a vertical releasing incision was made using blades #12 and #15. After full-thickness buccal flap elevation, an implant fixture (TSIII®; Osstem Implant Co., Seoul, South Korea; Superline®; Dentium, Seoul, Korea) was placed with adequate primary stability. After implant placement, GBR was performed using demineralized porcine bone mineral with 10% collagen (DPBM-C, LegoGraft®; Purgo, Seoul, Korea) and a resorbable collagen membrane (BioGide®; Geistlich Pharma AG, Wolhusen, Switzerland, or Ossix Plus®; Datum Dental Biotech, Telrad, Israel). The surgical site was sutured with 4-0 e-PTFE (Biotex®; Purgo Biologics, Seongnam, South Korea) and 5-0 nylon (Monosof®; Covidien, Dublin, Ireland) by using the modified horizontal mattress and interrupted suture methods. Stitch-out was performed two weeks after the operation, and reentry surgery was performed within four to six months after the implant surgery (Fig. 1).
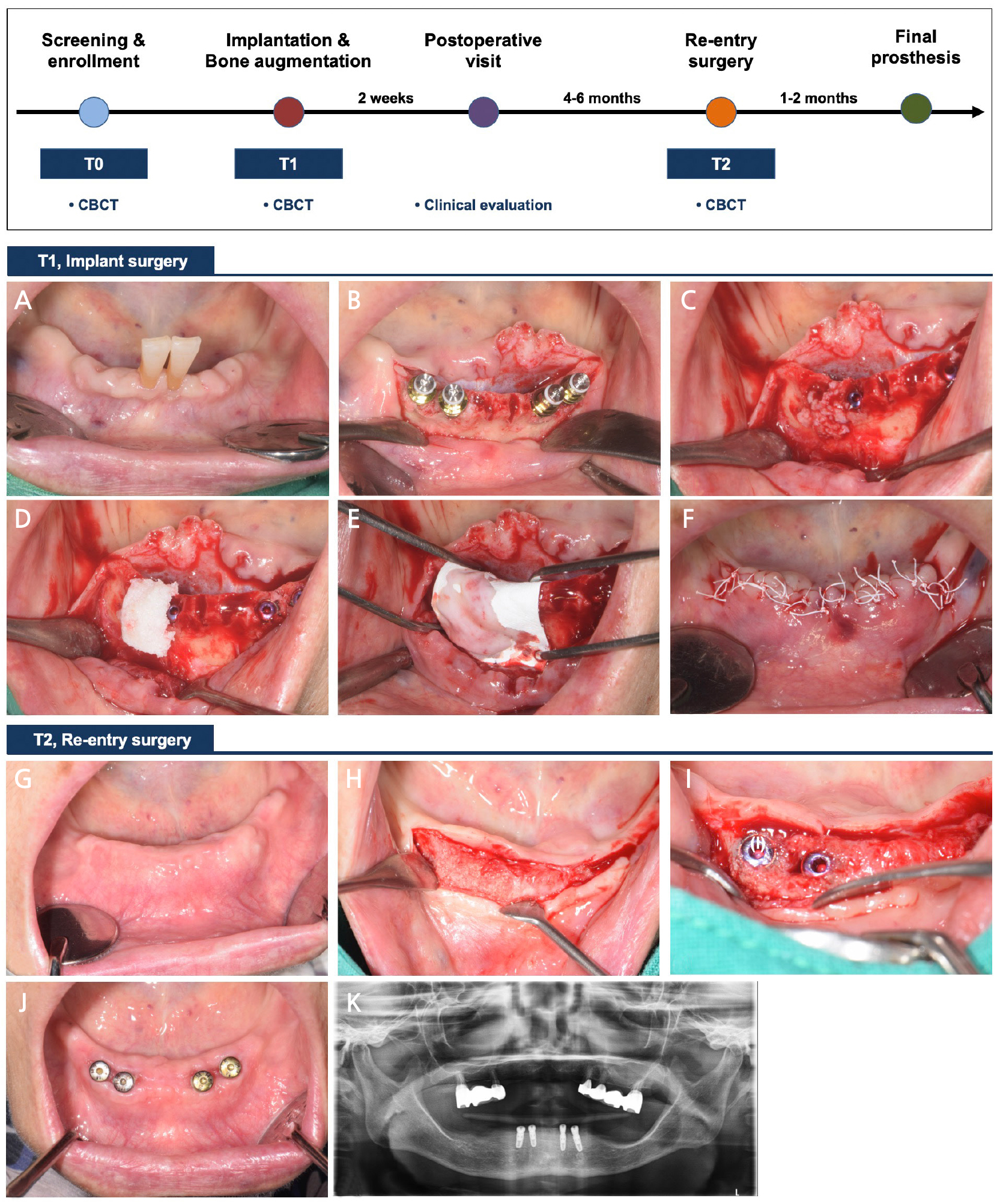
Fig. 1.
Schematic presentation of the treatment sequence and time points. (A–F) After dental implant placement, bone augmentation was performed with porcine-derived block bone substitutes and a resorbable collagen membrane. The flap was repositioned and sutured with primary closure, (G–I) At reentry, most of the peri-implant dehiscence defects were resolved, (J and K) Clinical and radiographic views after surgery.
3. Radiographic evaluation
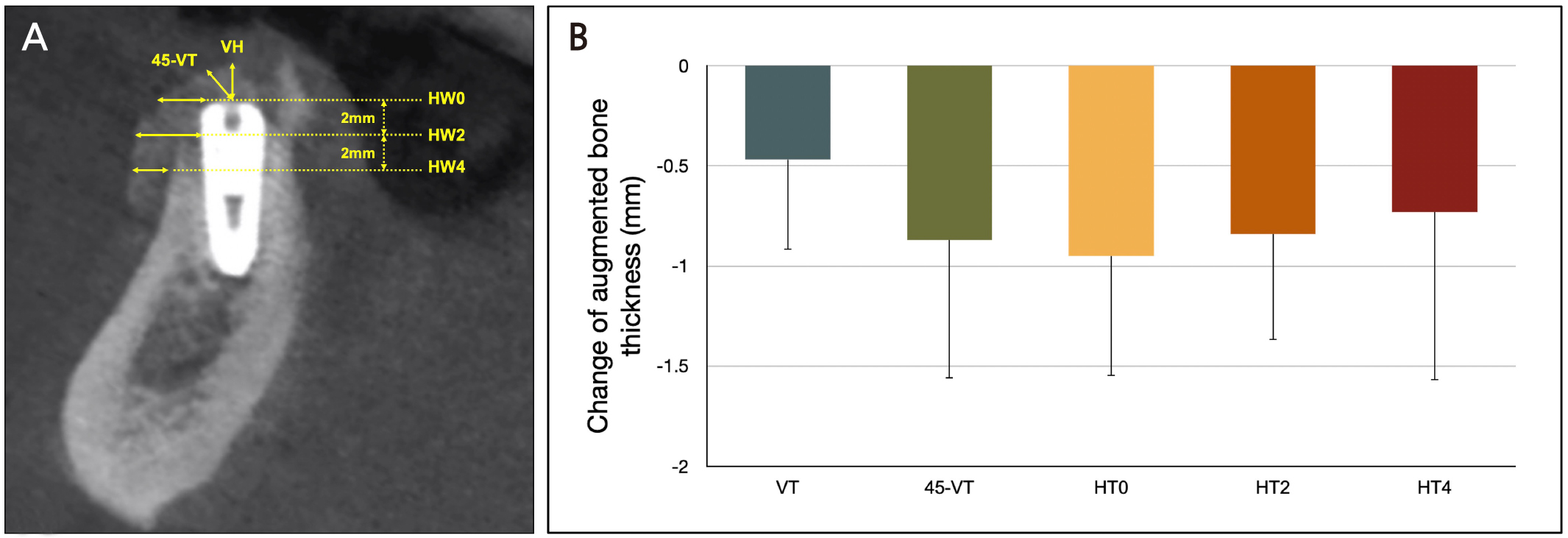
All subjects underwent cone-beam computed tomography immediately after implant placement (T1) and before reentry surgery (T2). Furthermore, the horizontal bone thickness was measured at the implant fixture shoulder (HW0) and apically at 2 (HW2) and 4 mm (HW4). Vertical bone thickness (VT) was measured along the long axis of the implant fixture from the implant shoulder to the most coronal level. The oblique bone thickness (45-VT) was measured at an angle of 45°, which divided the long axis and the perpendicular line drawn to this long axis (Fig. 2A). All radiographic parameters were assessed by a single calibrated examiner (JHL; intra-class correlation coefficient > 0.80).11
4. Self-reported questionnaire and clinical examination
The severity of subjective pain and swelling during the two weeks after implant surgery was evaluated using a visual analog scale (VAS) (transformed to a 0–10 scale). The duration of subjective pain and swelling was also assessed using a self-reported questionnaire. Early wound healing parameters, including wound dehiscence and membrane exposure, were evaluated by a single examiner (JHL) during the clinical examination.12
5. Statistical analysis
All clinical and radiographic outcomes were expressed as frequencies, proportions, means, standard deviations, medians, and first and third quartiles. The Shapiro–Wilk test was used to test for data normality, and the Wilcoxon signed rank test was used to determine the significance of the differences between T1 and T2. All calculations were performed using statistical software (Medcalc version 20.009; Mariakerke, Belgium), and p < .05 was considered statistically significant.
Ⅲ. Results
1. Baseline characteristics
A total of 12 patients (6 women and 6 men; mean age: 62.8 ± 8.9 years; age range: 51–81 years) with 20 surgical sites underwent implant surgery combined with GBR. Table 1 show a summary of the detailed characteristics of the patients and peri-implant dehiscence defects.
Table 1.
Baseline characteristics of patients and peri-implant dehiscence defects for 12 enrolled patients and 20 surgical sites
| Patient ID | Sex |
Age (years) |
Tooth position |
Diameter (mm) |
Length (mm) | Defect width (mm) | Defect height (mm) |
| 1 | M | 54 | #31 | 3.0 | 10.0 | 1.2 | 1.1 |
| #41 | 3.0 | 10.0 | 2.5 | 1.4 | |||
| 2 | M | 56 | #32 | 3.6 | 10.0 | 4.2 | 3.6 |
| 3 | M | 60 | #32 | 3.5 | 10.0 | 2.3 | 1.2 |
| #42 | 3.5 | 10.0 | 3.1 | 3.1 | |||
| 4 | F | 69 | #41 | 3.5 | 8.5 | 1.7 | 3.2 |
| 5 | M | 51 | #32 | 3.0 | 10.0 | 1.3 | 2.4 |
| 6 | F | 66 | #42 | 4.0 | 11.5 | 2.7 | 3.2 |
| 7 | F | 65 | #32 | 3.5 | 10.0 | 4.1 | 3.9 |
| 8 | M | 81 | #32 | 3.5 | 8.5 | 3.3 | 2.2 |
| 9 | F | 53 | #32 | 3.5 | 10.0 | 1.9 | 3.3 |
| #33 | 3.5 | 10.0 | 1.0 | 2.2 | |||
| #42 | 3.5 | 10.0 | 0.5 | 0.5 | |||
| #43 | 3.5 | 10.0 | 0.5 | 0.5 | |||
| 10 | M | 62 | #41 | 3.5 | 8.5 | 1.1 | 1.2 |
| 11 | F | 74 | #32 | 3.5 | 8.5 | 1.2 | 1.7 |
| #33 | 3.5 | 8.5 | 0.5 | 0.5 | |||
| #42 | 3.5 | 8.5 | 2.6 | 3.5 | |||
| #43 | 3.5 | 8.5 | 1.8 | 2.8 | |||
| 12 | F | 63 | #32 | 3.0 | 10.0 | 1.2 | 1.2 |
2. Radiographic outcomes
The vertical and horizontal augmented bone thickness in T1 was 2.17 ± 0.81 mm at VT, 3.39 ± 0.80 mm at 45-VT, 3.34 ± 0.83 mm at HT0, 3.35 ± 0.92 mm at HT2, and 2.55 ± 1.14 mm at HT4; the corresponding thickness in T2 was 1.70 ± 0.73 mm at VT, 1.89 ± 0.76 mm at 45-VT, 1.98 ± 0.72 mm at HT0, 1.89 ± 0.85 mm at HT2, and 1.39 ± 0.71 mm at HT4. There were significant differences between all time points (p < .05) (Table 2). The changes in vertical and horizontal bone thickness in T1–T2 was – 0.47 ± 0.45 mm at VT, – 0.87 ± 0.69 mm at 45-VT, – 0.95 ± 0.60 mm at HT0, – 0.84 ± 0.53 mm at HT2, and – 0.73 ± 0.84 mm at HT4 (Fig. 2B).
Table 2.
Changes in augmented bone thickness after GBR procedure of peri-implant dehiscence defects
| T1 | T2 | p-values | |
| VT (mm) |
2.17 ± 0.81 [2.15, (1.85, 2.79)] |
1.70 ± 0.73 [1.72, (1.17, 1.99)] | < .001 |
| 45-VT (mm) |
3.39 ± 0.80 [3.45, (3.14, 3.94)] |
1.89 ± 0.76 [2.12, (1.22, 2.42)] | < .001 |
| HT0 (mm) |
3.34 ± 0.83 [3.38, (3.00, 4.05)] |
1.98 ± 0.72 [2.19, (1.46, 2.47)] | < .001 |
| HT2 (mm) |
3.35 ± 0.92 [3.39, (2.89, 4.23)] |
1.89 ± 0.85 [2.00, (1.43, 2.29)] | < .001 |
| HT4 (mm) |
2.55 ± 1.14 [2.67, (1.86, 3.28)] |
1.39 ± 0.71 [1.22, (0.89, 2.03)] | .001 |
HT0, HT2, and HT4: Horizontal thickness at 0, 2, and 4 mm apically below the coronal end of the implant fixture.
3. Postoperative discomfort and wound healing outcomes
The severity of pain and swelling was 3.3 ± 2.5 and 3.9 ± 2.7 on the VAS, respectively, and the duration of pain and swelling was 3.7 ± 2.1 days and 6.1 ± 1.2 days, respectively. Among the enrolled patients, wound dehiscence and membrane exposure occurred at five surgical sites (25.0%; Bio-Gide®; n = 2 and Ossix Plus®; n = 3) (Table 3).
Ⅳ. Discussion
For predictive and successful GBR outcomes, space creation and maintenance are the major factors required to achieve sufficient bone regeneration.13 Although particulated xenogenic bone substitutes in combination with resorbable collagen membranes have been routinely used for GBR, these biomaterials are less efficient in providing space and stabilizing the wound.14 A number of different biomaterials (including pin, tenting screw, and titanium-reinforced membrane or mesh) have been proposed to prevent the collapse of the augmented area around the peri-implant dehiscence defects. However, the specific biomaterials that offer the best clinical outcomes are still unclear.15,16
Several studies have shown that the tight closure of a wound with sutures causes the significant loss of bone grafting materials, particularly the pronounced displacement in the occlusal–buccal space of the implant fixture shoulder.17,18 Recent in vitro cone-beam computed tomographic studies have compared the dimensional stability between particulated and block bone substitutes in combination with resorbable collagen membranes for the GBR of peri-implant defects.19 These studies show that there is significant displacement of the particulated demineralized bovine bone mineral (DBBM) during flap closure, which results in a partial collapse of the occlusal–buccal space. However, block-type DBBM exhibited less collapse than particulated DBBM. The amount of membrane collapse at the implant shoulder level was reduced by more than 50% (from –1.1 to – 0.5 mm) when block-type DBBM was used instead of particulated DBBM.
A recent study evaluated the augmentation stability of GBR in peri-implant dehiscence defects and reported that the change at HT0 and VT of the DBBM-C group (HT0: – 0.68 ± 0.53 mm [– 21.0%]; VT: – 0.91 ± 0.73 mm [– 30.1%]) was significantly more stable and persistent than that of particulated DBBM group (HT0: – 1.30 ± 0.77 mm [– 40.4%]; VT: – 1.57 ± 0.67 mm [– 52.0]; p < .05). 20 These results were consistent with those of the current study.
The study outcomes showed that there was a significant decrease in bone thickness after five months of implant surgery with GBR compared with the decrease immediately after surgery. However, bone gains of more than 1.5 mm were observed at all measurement sites compared with those at baseline.
Within the limitations of this study, only cases using DPBM-C could be included, and a direct comparison with particulated DPBM or DBBM-C could not be achieved. In addition, retrospective and short-term studies have inherent limitations. Further studies, including controlled clinical trials with long-term follow-up periods, are required.