Ⅰ. Introduction
Ⅱ. Patients and Methods
1. Patient selection and inclusion criteria
2. Data collection
3. Measurement of marginal bone loss with radiographs
4. Statistical analysis
Ⅲ. Results
1. Demographics and clinical outcomes
2. Implant survival rate
3. Implant success rate
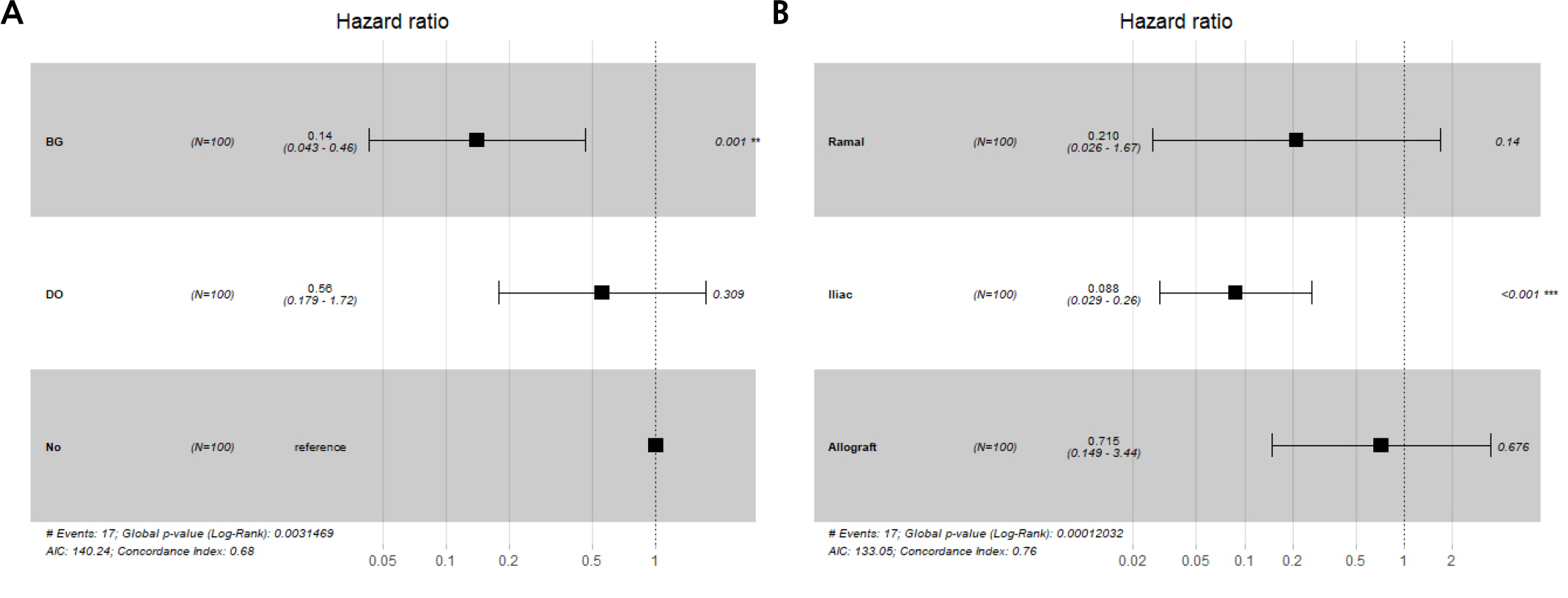
4. Cox proportional hazards ratios
Ⅳ. Discussion
Ⅴ. Conclusion
Ⅰ. Introduction
Mandibular defects result from various reasons such as tumors, trauma, and osteomyelitis.1 The use of vascularized bone flaps (VBF), including the scapula, fibula, and ilium, has been a reliable method for bone and adjacent soft tissue reconstruction for the past few decades.2 Mandibular reconstruction requires both the recovery of the mandible contour and occlusion.1 Adequate oral rehabilitation improves speech, masticatory function, and the quality of life.3
While non-vascularized bone grafts are appropriate for small defects, microvascular free flaps are typically used for the reconstruction of large bone defects.4 VBFs from the iliac and fibula are mainly used for the reconstruction of mandibular defects. Fibular flaps are suitable for reconstructing composite or simple long-span mandibular defects, and provides bone of sufficient quality and quantity for osseointegration of dental implants. Deep circumflex iliac artery flaps are suitable for reproducing the original contour of the jaw bone; the dimensions of the reconstructed bone are suitable for implant placement, which is advantageous for the dental rehabilitation and aesthetic reconstruction for mandibular defects.4 Additionally, vertical distraction osteogenesis can reduce vertical discrepancies resulting from VBF for mandibular reconstruction.5
Oral rehabilitation after mandibular reconstruction is performed either using conventional dentures or by implant placement. Owing to the unfavorable oral environment common after mandibular reconstruction, such as insufficient bone height, changes in the soft tissue environment, and loss of sensation, conventional dentures may be ineffective in restoring masticatory function. Implant-supported fixed prostheses or overdentures can be better alternatives for rehabilitating masticatory function and present some advantages over conventional dentures, including a stronger masticatory force and good aesthetics.6 However, they may also present some disadvantages. The grafted tissues are much thicker than the gingiva, not fixed to the bone by the periosteum, and not keratinized.7 Furthermore, the anatomical difference between the flap and the mandible results in a vertical discrepancy, increases the crown-fixture length ratio, and endangers implant stability.7
Currently, many studies have reported the survival and success rates of implants placed in VBFs. However, there is a lack consensus on the criteria for success rate, and only a few studies have objectively evaluated success rates through radiological evaluations. Therefore, this study aimed to evaluate the marginal bone loss, survival, and success rate of implants placed in reconstructed mandibles using a VBF and to identify prognostic factors that may influence bone loss.
Ⅱ. Patients and Methods
1. Patient selection and inclusion criteria
The study protocol was reviewed and approved by the Institutional Review Board of Chonnam National University Hospital (IRB no. CNUH-2022-163). This retrospective study included patients who underwent mandibular reconstruction surgery using VBF followed by delayed implant placement at the Department of Oral and Maxillofacial Surgery, Chonnam National University Hospital, between January 2007 and December 2019. Additional inclusion criteria were a follow-up period of more than one year and panoramic or periapical radiographs examinations.
2. Data collection
The following patient data were extracted: age, sex, diagnosis, reconstruction method, number of failed or failing implants, radiation therapy or chemotherapy, tumor site, resection range, reconstruction plate removal period, additional bone graft, implant placement period, implant system, width, length, location, type of implant prosthesis, related complications, smoking habits, medical history, marginal bone loss, and period (Table 1).
3. Measurement of marginal bone loss with radiographs
The criteria for failing implants was set at a vertical bone loss below 1 mm during the first year and 0.2 mm for each subsequent year, as proposed by Albrektsson et al.8 Marginal bone loss (MBL) was measured by comparing the panoramic or periapical radiographs taken at different time points. The number and period of failing implants were evaluated based on the time and length at which abnormal MBLs were detected. MBL was measured using the length measurement tool in INFINITT PACS M6 (Infinitt Healthcare, Seoul, Korea). After measuring the fixture length on radiographs, the magnification ratio (MR) was calculated by dividing the value by the actual fixture length. The distance from the crestal bone, with marginal bone loss, to the upper part of the fixture was measured. Finally, this value was divided according to the MR. The measured MBL values were rounded to two decimal places. (Fig. 1).
4. Statistical analysis
All statistical analyses were performed using RStudio (version 4.2.1; RStudio Inc., Boston, MA, USA), and the survival and success rate curves of the implants were analyzed using a Kaplan-Meier estimation and Cox proportional hazards model.
Table 1.
Summary of patient’s data evaluating the survival and success rate of implants placed in VBF
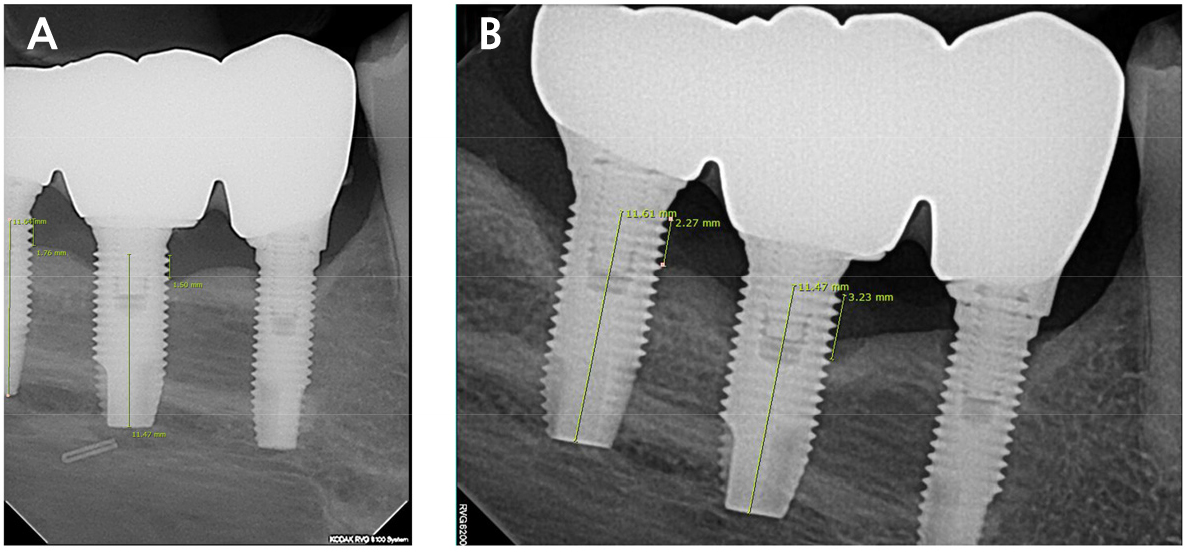
Fig. 1.
Measurement of MBL and implant length. The actual fixture length of #46i and #47i were both 11.5 mm. (A) Periapical radiograph 1 year after implant placement, (M.R #46i: 0.997, #47i: 1.012) (B) Periapical radiograph 3 years after implant placement. (M.R #46i: 0.997, #47i: 1.009), As for MBL, #46i increased from 1.5 mm to 3.2 mm, and #47i increased from 1.7 mm to 2.2 mm.
Ⅲ. Results
1. Demographics and clinical outcomes
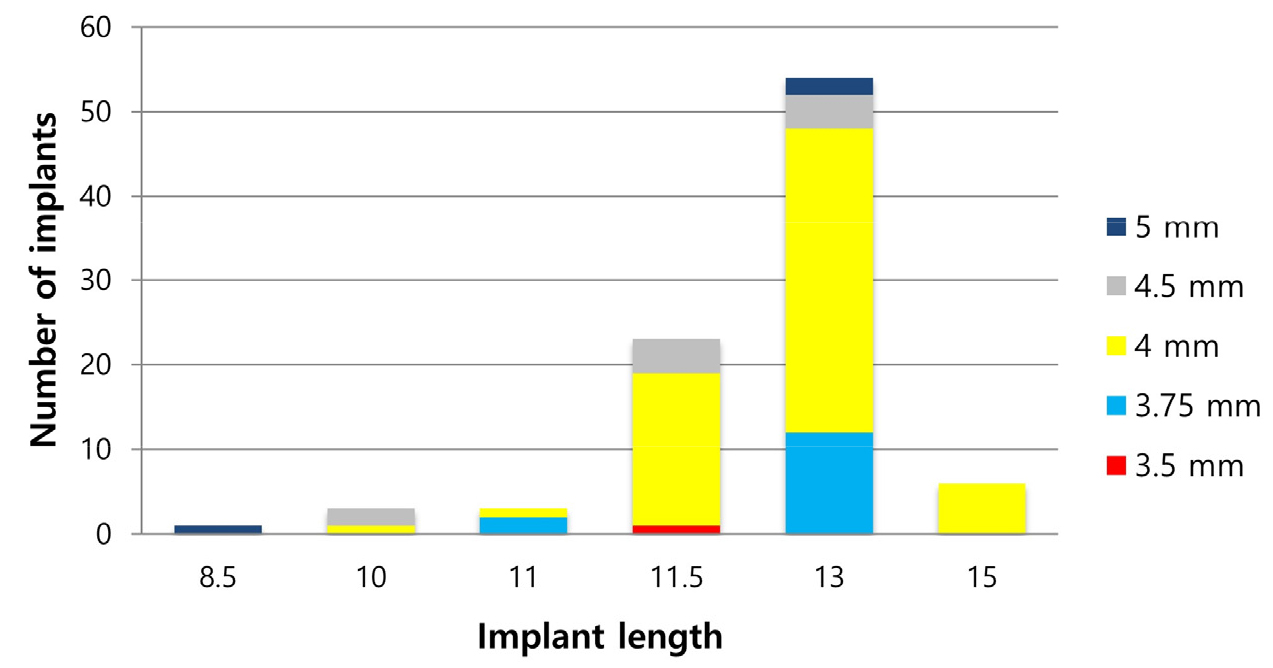
From January 2007 to December 2019, 80 patients underwent mandibular reconstruction surgery using VBF. Twenty-six patients underwent delayed implant placements, of which five were excluded because of a lack of radiographic follow-up data for more than one year. Therefore, this retrospective study finally included the medical records of 21 patients with 100 implants. All implants were of the external connection type (USII; Osstem®, Seoul, Korea) and placed at the bone level. The patients’ mean age was 49.7 years (14−75 years). This study included 11 male and 10 female patients. Fibula and iliac bone flaps were used for 16 and 5 patients, respectively. Osteomyocutaneous flaps were used in 13 patients, and myo-osseous flaps followed by direct closure in eight patients. Reconstruction plate removal was performed after an average of 10.6 months (5−18 months). Delayed implant placement was performed at an average of 16.7 months (7−45 months) after mandibular reconstructive surgery. The mean follow-up period after implant placement was 80.6 months (12−144 months). Additional bone grafting was performed in 17 patients. Iliac block bone or particulate marrow cancellous bone grafts and allografts (OraGRAFT®; LifeNet Health, Virginia beach, VA, USA) were used in 14 and 3 patients, respectively. Vertical distraction osteogenesis (DO) was performed in four patients, of which three underwent DO surgery using an iliac block bone graft and one with an ostectomy of the fibula bone itself without bone graft. DO surgery was performed an average of 20.7 months (7−34 months) after the mandibular reconstruction surgery. The mean distraction period was six months (4−7 months) and the mean height of the distracted bone was 10 mm (6−15 mm). Four patients showed bone exposure, two had local recurrence, and 15 had no significant complications. The length and diameter of the implants are shown in Fig. 2.
2. Implant survival rate
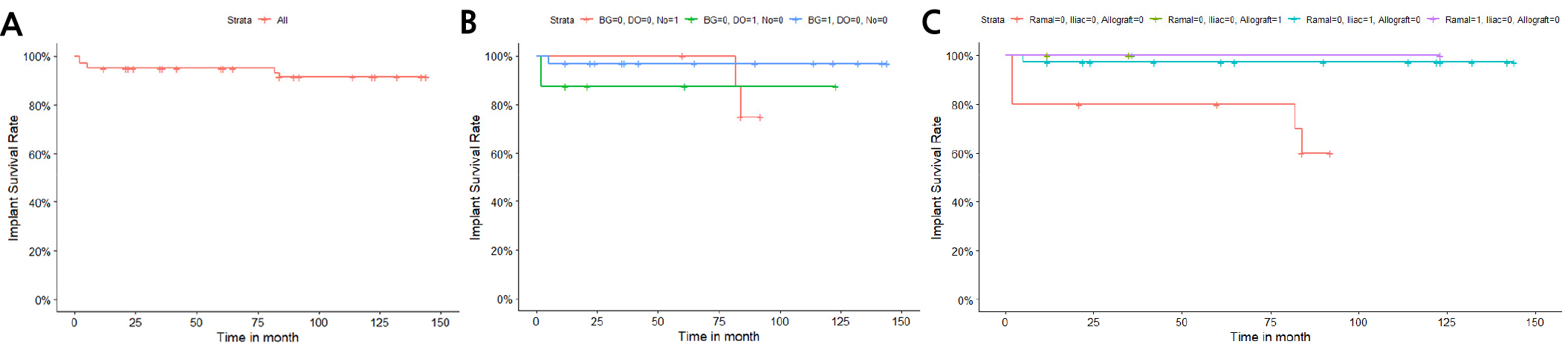
Seven implants in three patients failed, all of which had bone exposure complications (Table 2). Fig. 3 shows a typical case of implant success. The 5- and 10-year implant survival rates were 95.0% and 91.5%, respectively (Fig. 4A). When only a bone graft was performed, the survival rate of implants was 96.9%; for DO with or without a bone graft, the survival rate was 87.5%; and with neither, the survival rate was 75% (Fig. 4B). When bone grafting was not performed or only DO was performed, the survival rate of implants was 60%. The survival rate of implants was 97.3% for iliac bone grafts. (Fig. 4C).
Table 2.
An overview of failed implants
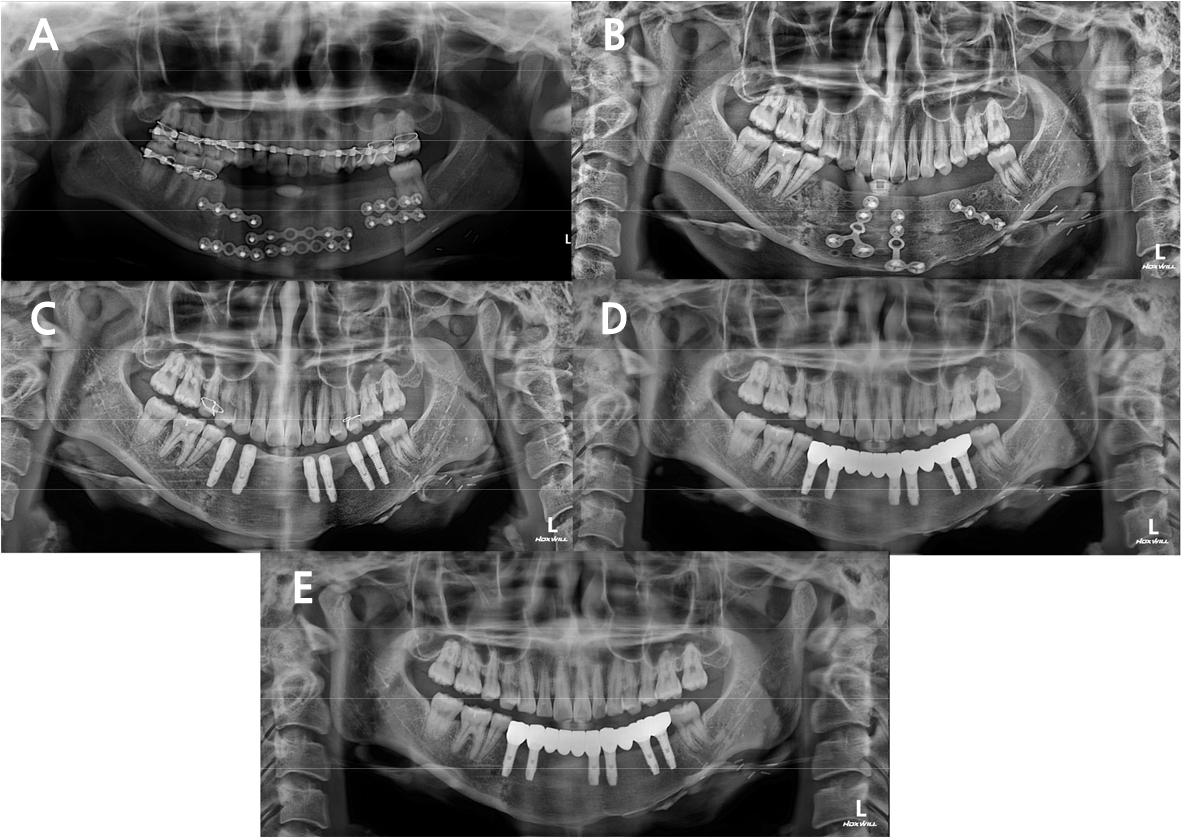
Fig. 3.
(A) Due to the recurred odontogenic keratocyst in the mandible, the patient underwent segmental mandibulectomy and iliac bone flap surgery, (B) 14 months later, additional iliac block bone graft was performed, (C) 7 months after the additional bone graft, delayed implant placement was performed, (D) 4 months after the implant placement, the temporary prosthesis was delivered. (E) 2 years after the implant placement, panoramic radiograph was taken. No significant MBL or symptoms were found.
3. Implant success rate
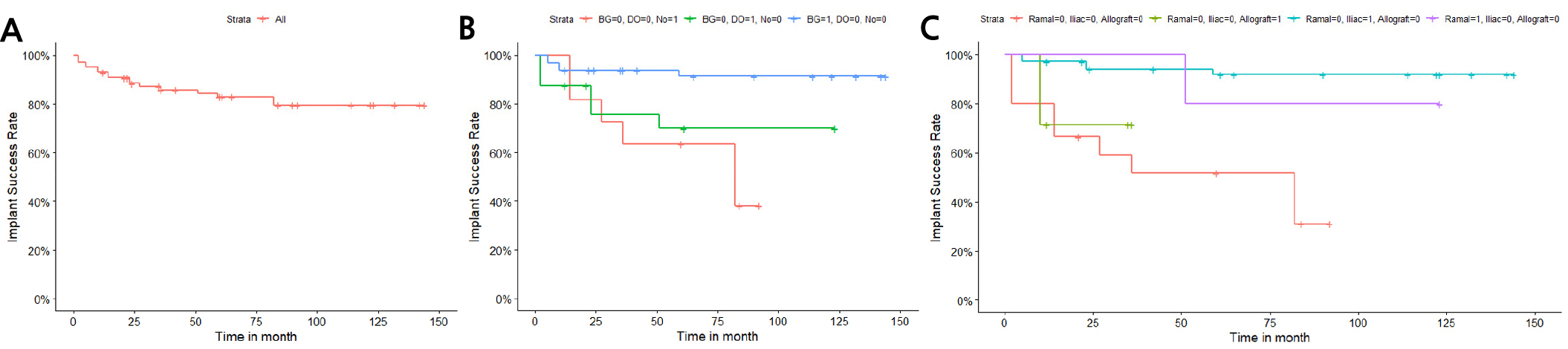
Of the 100 implants, 7 failed, 28 showed MBL, and 65 showed no specific MBL. Implants showing MBL are summarized in Table 3. Ten implants showed an abnormal MBL. The mean MBL of the 28 implants with MBL was approximately 1.9 mm (0.5−5.1 mm), and 3.1 mm (1.3−5.1 mm) for the 10 implants with abnormal MBL. The 5- and 10-year implant success rates were 82.8% and 79.5%, respectively (Fig. 5A). For bone grafting only, the implant success rate was 91.4%; for DO with or without bone graft, the success rate was 70.0%; and for neither, the success rate was 38.2% (Fig. 5B). When bone grafting was not performed or DO was performed only, the success rate was 31.1%. The success rates were 92.0%, 80.0%, and 71.4% for iliac bone grafts, ramal bone grafts, and allografts, respectively (Fig. 5C).
Table 3.
An overview of implants showing MBL (marginal bone loss)
4. Cox proportional hazards ratios
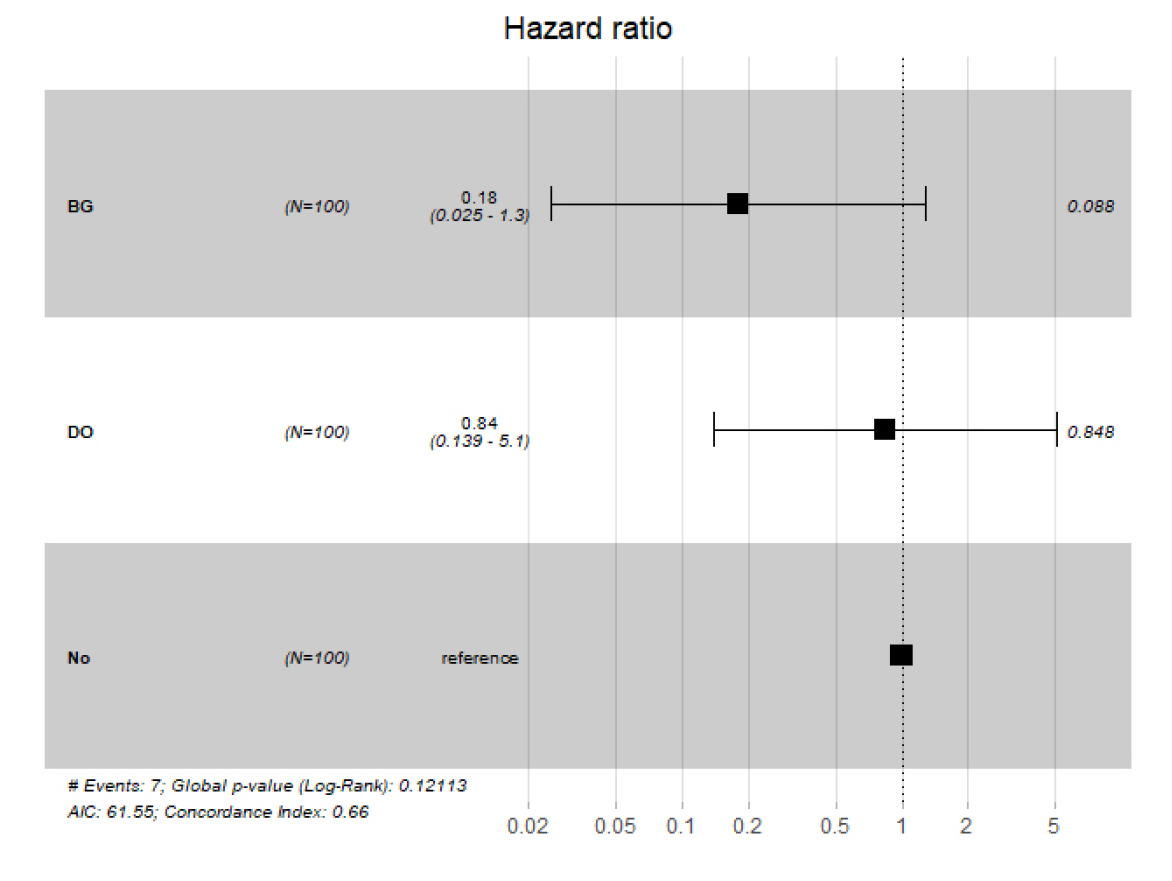
The hazard ratios of implant survival were 0.18 and 0.84 with bone grafting and DO surgery, respectively, although neither were statistically significant (Fig. 6). The hazard ratio of implant success was 0.14 with bone graft (statistically significant; p < .05) and 0.56 with DO surgery (not statistically significant) (Fig. 7A). This suggests a lower risk of implant failure when additional bone grafts are used. Moreover, the hazard ratio of implant success was 0.088 for iliac bone grafts (statistically significant; p < .05), and had a higher implant success rate than the other materials (Fig. 7B).
Ⅳ. Discussion
The osseointegration of implants into a fibula flap is safe and predictable, with a prognosis similar to that of a natural mandible.9 Lei et al. reported a 93.2% cumulative survival rate of implants placed in bone flaps, with a longest follow-up of 12.9 years;10 Wei et al. reported 5-, 10-, and 20-year implant survival rates of 90.1%, 83.1%, and 69.3%, respectively;11 Gerardo et al. reported 1-, 5-, and 10-year implant survival rates of 97.2%, 86.5%, and 79.3%, respectively;12 Sameh et al. reported 1- and 11-year cumulative survival rates of 93% and 78%, respectively;13 and Hongyang et al. reported 1-, 2- and 5-year cumulative survival rates of 96%, 87%, and 81%, respectively.14 In this study, only 7 out of 100 implants failed, with 5- and 10-year implant survival rates of 95.0% and 91.5%, respectively. Interestingly, all seven failed implants were from the only three patients with complication of bone exposure. Complication such as bone exposure after mandibular reconstruction surgery could make the mandible vulnerable to infection and cause osteomyelitis, which may affect implant osseointegration through changes in the bone and surrounding soft tissues. Furthermore, three patients with 20 implants underwent DO with iliac block bone grafts, and one patient with four implants underwent DO without additional bone grafting. Among these, five implants failed and three showed abnormal MBL. Three failed implants were in patients who underwent DO without additional bone grafting. The Kaplan-Meier survival curves according to bone graft, DO, and bone graft material (Fig. 4B, 4C) shows a higher implant survival rate for the group that underwent only bone grafting. Although the number of ramal bone and allograft cases was small, iliac bone grafts had a relatively high survival rate of 97.3%. There were no statistically significant differences in the Cox proportional hazard ratios. However, additional bone grafting before implant placement resolved the vertical discrepancy, lowered the crown-fixture length ratio, and subsequently had a positive effect on the implant prognoses.
Most studies have only reported implant survival rates, with heterogeneities in the definition of implant success rate.15 Success rate has mostly been defined as the absence of implant-related pain, suppuration, swelling, mobility, discomfort, ongoing pathological processes, peri-implantitis, neuropathies, or persistent paresthesia, as noted by van Steenbergher.16 Fatih et al. reported a fibula flap implant success rate of 92.6%;17 Maria et al. reported a 93% implant success rate;18 and Gerardo et al. reported 1-, 5-, and 10-year implant success rates of 95.4%, 73.5%, and 64.7%, respectively.12 One systematic review of 910 implants placed in vascularized fibular grafts determined a 40-month success rate of 92.6% (82−100%).19 Implants placed in fibula flaps had an estimated 94% success rate (95% CI [confidence interval] = 0.91 to 0.96]) with an insignificant heterogeneity of 37%.20 In this study, the success rate was determined by measuring the MBL through panoramic or periapical radiographs using Albrektsson’s criteria.8 To visually and objectively evaluate implant success rates, we hypothesized that measuring and evaluating the MBL would be more accurate. In this study, 10 implants in seven patients showed abnormal MBL, all of which were placed in the fibula flap. However, the mean MBL of 10 failing implants was 3.1 mm and was not clinically significant. The 10 implants in seven patients who showed abnormal MBL, included five implants in four patients who did not undergo additional bone grafting. As seen in the Kaplan-Meier survival curves according to bone graft, DO, and bone graft material (Fig. 5B, 5C), the implant success rate was higher in the group that only received bone graft, and the iliac bone grafts had a high success rate (92.0%) than other materials. Statistically significant Cox proportional hazards ratios were obtained for the bone graft and iliac bone graft groups. Therefore, reducing vertical discrepancy and crown-fixture length ratio through an additional iliac bone graft could be an important factor in determining the success of implants placed in VBFs. In the case of implants with abnormal MBL observed in this study, the risk factors that could be considered are the presence of complications, such as bone exposure and lack of additional bone grafting.
The optimal period between mandibular reconstruction and implant placement remains controversial. Urken et al. were the first to propose rehabilitation with simultaneous reconstruction and implantation.21 De Santis et al. proposed a two-step protocol with mandibular reconstruction and implant placement at different stages (with a six-month waiting period).22 Maria et al. suggested that the presence of immediate dental implants at the time of fibula transfer neither delays radiotherapy nor increases acute radiotherapy treatment toxicities.18 Panchal et al. reported a higher survival rate for immediate implant placement (97.0%) than for delayed placement (89.9%), with median follow-ups of 14 and 40 months, respectively.23 However, Bodard et al. argued that immediate implant placement after mandibular reconstruction may damage bone viability, increase operation time, and result in implant malposition, thereby considerably increasing the period of ischemia and the risk of non-integration.24, 25 Moreover, placement at the same time as reconstruction is technically difficult as implant placement, flap placement, prosthetic result, and soft tissues must all be taken into account.7 Our department preferred delayed implant placement due to the possibility of recurrence, high cost, unstable bone viability, extended operation time, and difficulty in ideal placement for simultaneous implantation. Bodard et al. suggested that delayed implant placements should be performed at least 6−12 months after the graft, when the bone remodeling and muscle healing are complete.7 We recommend delayed implant placement approximately six months after mandibular reconstruction surgery using VBF; all implants in this study were placed after an average of 16.7 (7−27) months, with favorable prognoses.
There are several limitations to implant placement after mandibular reconstruction surgery using VBF. The soft tissues of VBF are much thicker than the gingiva, and are not fixed to the bone by the periosteum or keratinized.7 Additionally, a fibula flap placed only at the inferior border of the mandible can result in a vertical discrepancy between the graft segment and occlusal plane which in turn adversely affects implant mechanics or denture stability and retention.26 Lee et al. reported a crown-fixture length ratio of approximately 1:1.21 for the conventional one-strut method, which is higher than the 1:1.5 ratio recommended for patients with normal alveolar and basal portions of the mandible and may adversely affect the implant mechanics or denture stability and retention.26 Reychler et al. observed abnormal occlusal relationships between the maxilla and mandible after mandibular reconstruction, a vertical discrepancy between grafted and nongrafted zones, and a lack of vestibule and granular soft tissue at the implant recipient site, caused by a lack of keratinized tissue.27 Conversely, Bodard et al. reported satisfactory results for implant-supported prosthesis after mandibular reconstruction despite the thickness and mobility of soft tissues, scar contracture, and the absence of keratinization, regardless of radiation.24 Due to the cumulative effects of radiation on bone vascularity, the regenerative capacity of these tissues is limited, which may be detrimental to subsequent implant osseointegration.28 However, the effect of radiotherapy on implants remains controversial. Fundamental changes in implant survival in patients receiving radiation have resulted from recent advancements in implant therapy, including three-dimensional planning, guided implant surgery, technical improvements in implant surface features, and revised treatment concepts. Thus, dental implants now seem to be a favorable treatment option for the oral rehabilitation of patients with head and neck cancer after radiotherapy.29 Moreover, Granstrom et al. reported no general consensus on whether implant failure occurs more frequently in patients who have undergone radiotherapy.30 However, the radiation dose must be considered: soft tissue necrosis can result from doses < 50 Gy and salivary gland injuries can occur after doses < 20 Gy.29 The risk and severity of osteoradionecrosis is related to the radiation dose, volume of irradiated tissue, and patient’s dental health.31 Therefore, it is essential to obtain radiation dose distribution data from radiation oncologists to determine the best locations for implant insertion.32 In this study, since only one patient received radiotherapy, we were unable to assess radiotherapy as a risk factor for implant survival.
The limitations of this study were the follow-up period variation of 12−144 months and the relatively small number of cases. Although all failed implants occurred in the fibula flap, the number of cases was too small to draw definite conclusions; future studies on the differences between the fibula and iliac bone flaps will be necessary. Another limitation was that patients’ symptoms, such as bleeding on probing, were improperly characterized. As a result, peri-implantitis could not be evaluated, and the implant success rate was calculated by measuring MBL using radiographs in our study. Simple radiographic evaluations alone have limitations in evaluating the success rate of implants. In this study, some patients did not have periapical radiographs and only had panoramic radiographs, which may have affected the accuracy of MBL measurement. In addition, there will have been some changes in implant surface treatment over the 13-year period. However, it is important that the MBL measurements and implant success rate calculations were performed objectively through radiographic evaluation. Furthermore, since all implants were of the external connection type, differences in connection type and insertion depth had no significant effect on the result.
Despite these limitations, the survival and success rates of all 100 implants in our retrospective study were satisfactory, and reconstruction of the mandible through VBF and oral rehabilitation through delayed implant placement were considered reliable and successful methods. Moreover, had the implants been placed in ideal locations using implant guide surgery, this study may have shown a better prognosis of implants placed in the VBF.
Ⅴ. Conclusion
Reconstruction of the mandible using VBF and oral rehabilitation through delayed implant placement are useful and reliable methods to restore masticatory function and improve quality of life and socioemotional problems. Complications such as bone exposure and osteomyelitis and undergoing additional bone grafting are risk factors for implant failure.