Ⅰ. Introduction
Ⅱ. Case Report
1. Patient History and Initial Presentation
2. Clinical and Radiographic Examination
3. Surgical Procedure
4. Healing Period and Implant Placement
5. Prosthetic Rehabilitation
6. Follow-up
Ⅲ. Discussion
Ⅰ. Introduction
Chemical burns in the oral cavity can result in immediate tissue damage and long-term complications, including alveolar bone loss, root resorption, and structural defects.1 Such injuries can cause severe tissue necrosis and delayed sequelae that may not be clinically apparent for several years.2
Reconstruction of segmental alveolar bone defects in the aesthetic zone presents a unique challenge. Unlike contained defects, these defects lack one or more bony walls, making it difficult to achieve and maintain volume stability.3 The anterior maxilla is particularly challenging because of a thin buccal bone, high aesthetic demand, and the need for adequate bone volume for implant placement.4
Although various bone grafting materials have been introduced including allografts, xenografts, and alloplastic materials, autogenous bone remains the gold standard for predictable bone regeneration, particularly for significant defects.5,6 Ramal cortical bone offers several advantages such as the ability to be harvested intraorally under local anesthesia, excellent structural support, and superior volume stability compared with particulate grafts alone.7
The use of local soft tissue as a biological membrane has been reported to enhance bone regeneration by providing vascularization and containing graft materials.8 In cases where healthy palatal tissue overlies a bony defect, using this tissue as a flap may provide an ideal biological environment for bone healing.
This case report describes successful reconstruction of a maxillary segmental bony defect using ramal cortical bone and an allogenic bone graft. This was followed by implant-supported prosthetic rehabilitation with complete bone regeneration maintained over a 3-year follow-up period.
Ⅱ. Case Report
1. Patient History and Initial Presentation
A male patient in his 50s age group presented with a chief complaint of food impaction in the right maxillary palatal area. The patient's history showed that he had experienced chemical burns from sulfuric acid exposure approximately 40 years before presentation, which resulted in chemical burns in the oral cavity and palate. The patient stated that, following the initial injury, he experienced palatal tissue damage but had not seeked specific treatment. The area remained asymptomatic for decades until he noticed food accumulation in the palatal region.
2. Clinical and Radiographic Examination
Clinical examination revealed a palatal segmental bony defect in the right maxillary region covered by healthy-appearing gingival tissue. The defect communicated between the oral cavity and the palatal vault. Multiple teeth in the right maxillary quadrant exhibited increased mobility and displacement.
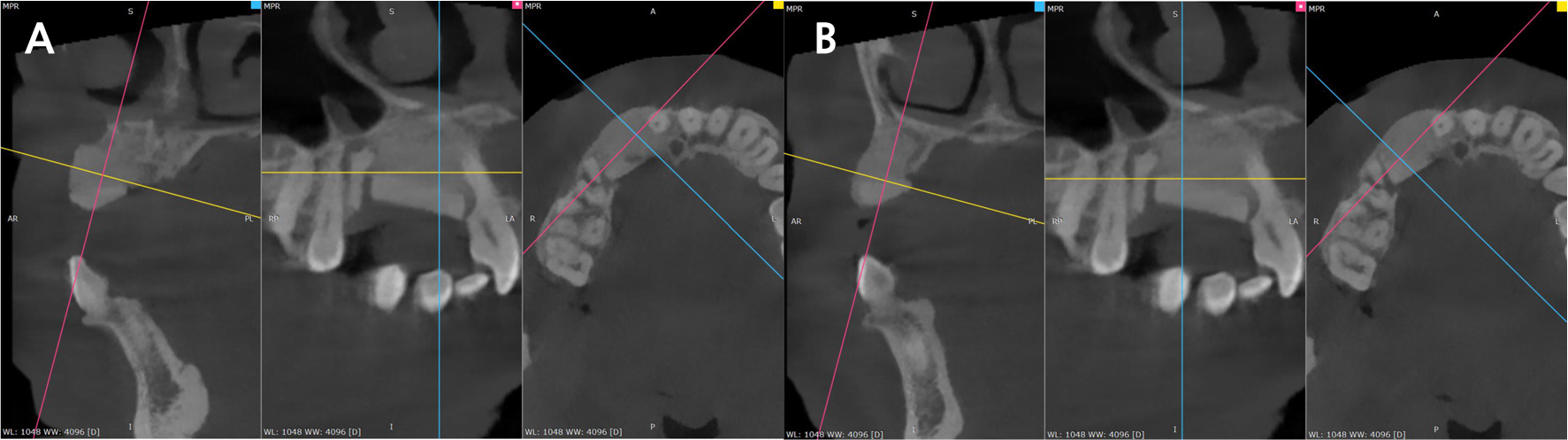
Panoramic radiography and cone-beam computed tomography (CBCT) revealed multiple external root resorption involving teeth #11, #12, #13, #14, and #15 (Fig. 1A‑C). An extensive segmental bone defect was evident in the right maxillary alveolus, extending from the palatal to the buccal cortex, with a significant loss of both alveolar bone height and width in the affected area (Fig. 1D). Notably, the defective area showed no evidence of osteolytic lesions, cysts, or other pathological findings.
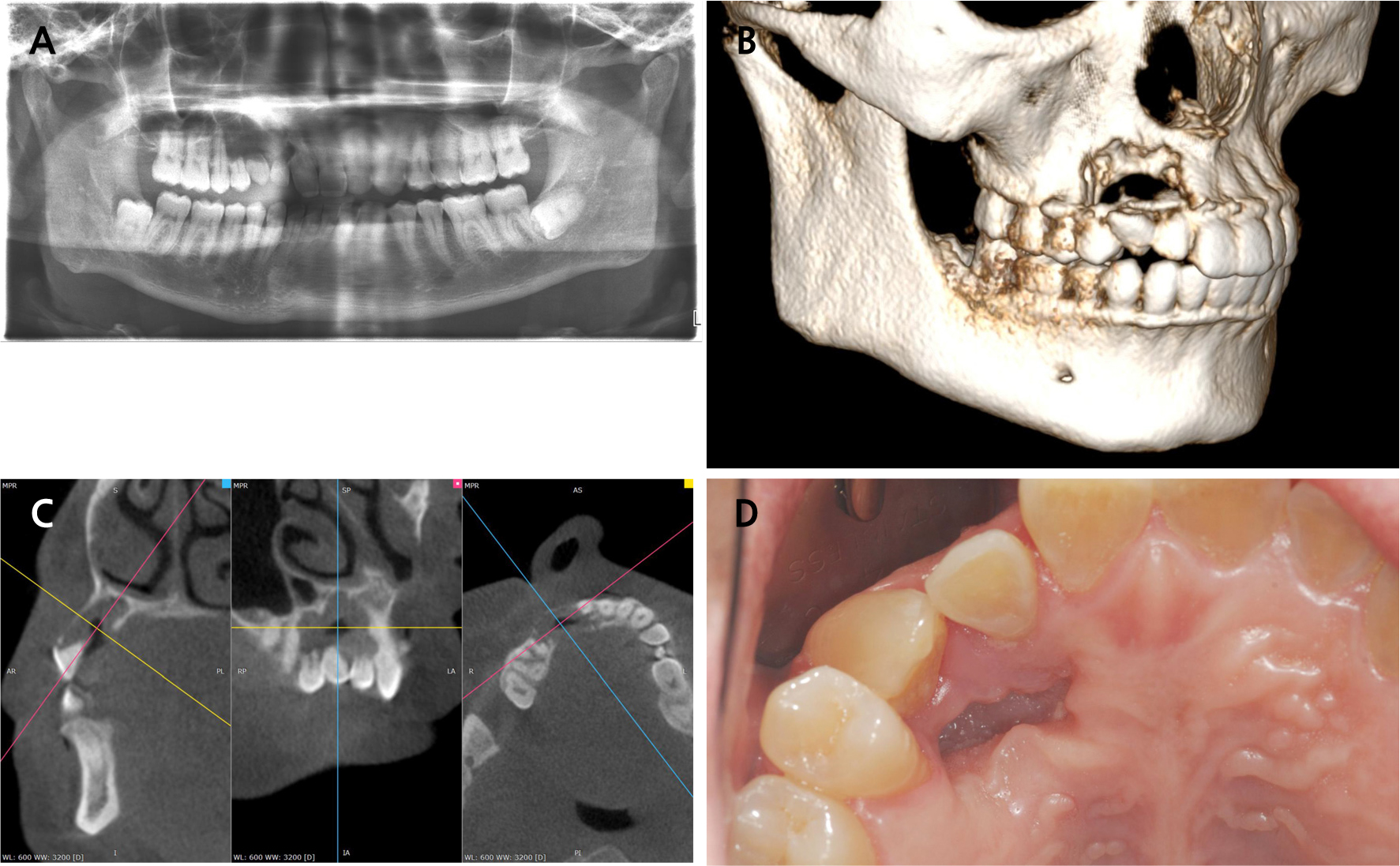
Fig. 1.
Initial presentation. (A) Panoramic radiograph showing multiple root resorption and extensive bone defect in the right maxilla, (B) Three-dimensional CBCT reconstruction demonstrating segmental bone defect, (C) Cross-sectional CBCT images showing the extent of palatal bone loss and defect morphology, (D) Intraoral photograph showing the palatal defect covered by healthy gingival tissue.
Based on the clinical and radiographic findings, teeth #12, #13, and #14 were deemed hopeless because of severe root resorption and lack of bony support. The treatment plan includedextraction of these teeth and bone reconstruction using autogenous and allogeneic bone grafts followed by implant placement and fixed prosthetic rehabilitation.
3. Surgical Procedure
Under general anesthesia, teeth #12, 13, and 14 were extracted atraumatically. The palatal soft tissue covering the bony defect was carefully elevated using a full-thickness mucoperiosteal flap. Intraoperatively, the bone defect was confirmed to be a segmental bony defect with loss of both the buccal and palatal cortical plates (Fig. 2A). No pathological soft tissues or abnormal findings were noted within the defect area, which was lined with healthy gingival epithelium.
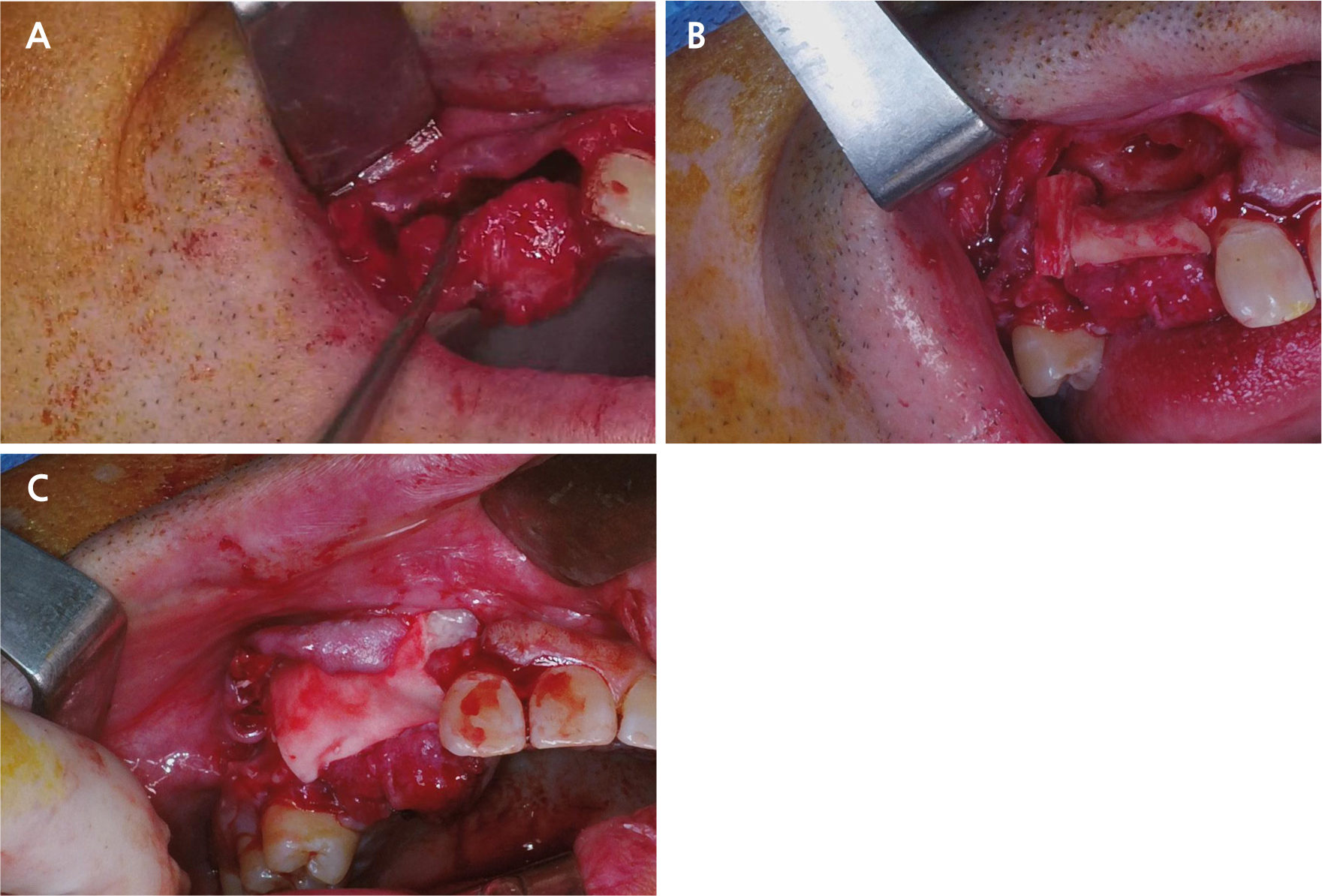
Fig. 2.
Surgical procedure. (A) Clinical view of the segmental bone defect after atraumatic extraction of teeth #12, #13, and #14, (B) Bone graft placement of harvested cortical bone block from the external oblique ridge of the right mandibular ramus filling the defect space, (C) Resorbable membrane placement and tension-free primary closure achieved using the elevated palatal flap.
The defect was reconstructed using the following protocol. A cortical bone block was harvested from the external oblique ridge of the right mandibular ramus and adapted to provide structural support through mechanical fixation without a plate or screw (Fig. 2B). Particulate allogenic bone graft (SureOss Chip; HansBiomed Corporation, Seoul, Korea) was then placed to fill the remaining defect space, and fibrin sealant (Tisseal; Baxter Healthcare Corporation, Deerfield, IL, USA) was applied to stabilize the graft materials. A resorbable collagen membrane (OVIS membrane; Dentis Co., Daegu, Korea) was positioned over the grafted area, followed by application of platelet-rich fibrin (PRF). Finally, a tension-free primary closure was achieved using an elevated palatal flap as the covering tissue (Fig. 2C). The patient was prescribed postoperative antibiotics and analgesics and instructed on postoperative care of soft diet and oral hygiene maintenance.
4. Healing Period and Implant Placement
The patient reported temporary numbness of the right lower lip area and a sensation of reduced oral volume following surgery, which gradually resolved. At the 2-week follow-up, the surgical site showed uneventful healing, with no signs of infection or dehiscence.
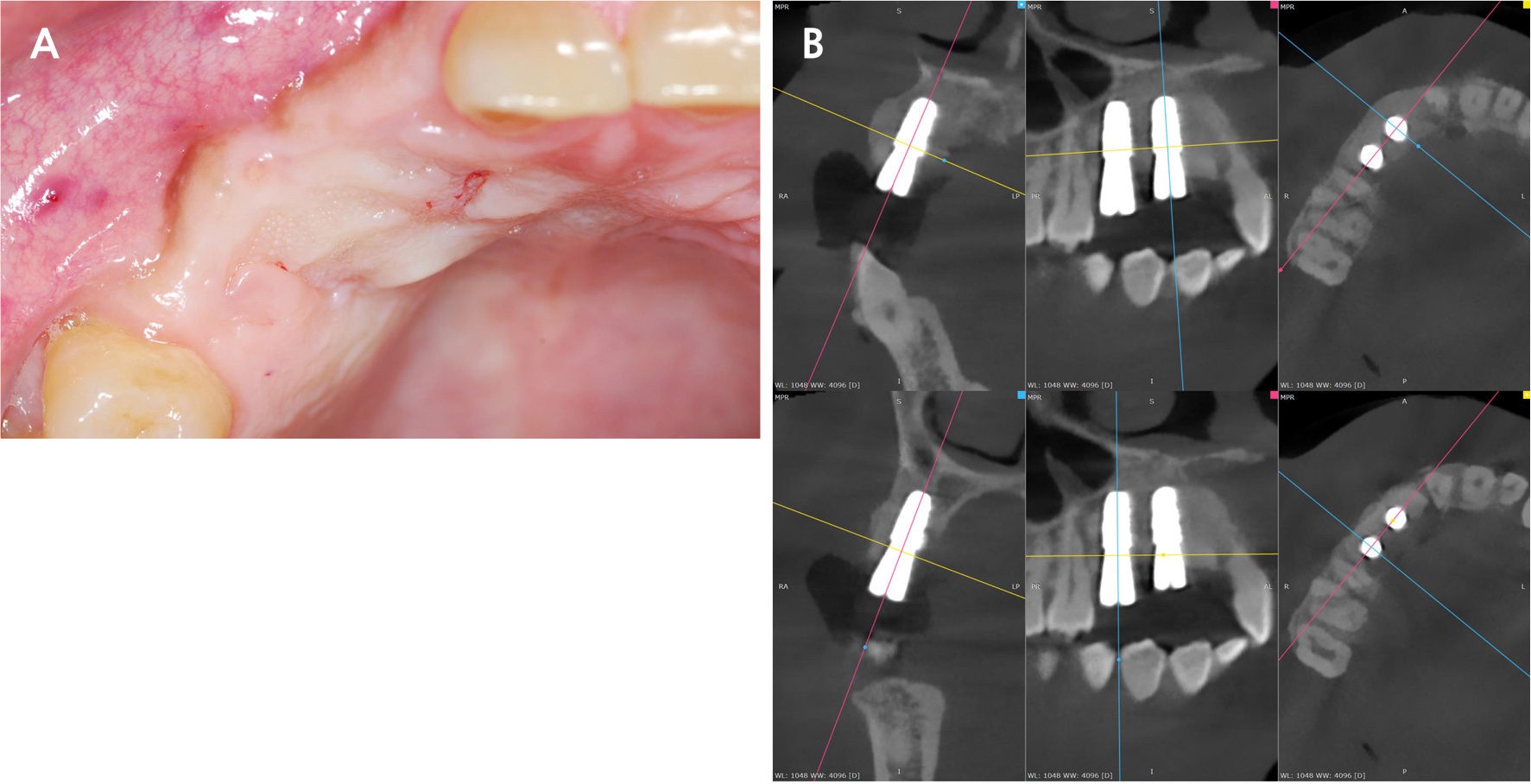
Six months after grafting, CBCT evaluation demonstrated excellent bone regeneration with complete incorporation of the graft materials and formation of mature bone in the previously defective area. This site was deemed suitable for implant placement (Fig. 3A and 3B).
Guided implant surgery was planned by merging the CBCT and intraoral scan data. A surgical guide was fabricated to accommodate the prosthetic design. Two dental implants (TS III BA, 4.5 mm diameter × 8.5 mm length; Osstem, Seoul, Korea) were placed at positions #13 and #14 under local anesthesia (Fig. 4A and 4B). Primary stability exceeding 30 N was achieved for both implants and healing abutments were connected.
5. Prosthetic Rehabilitation
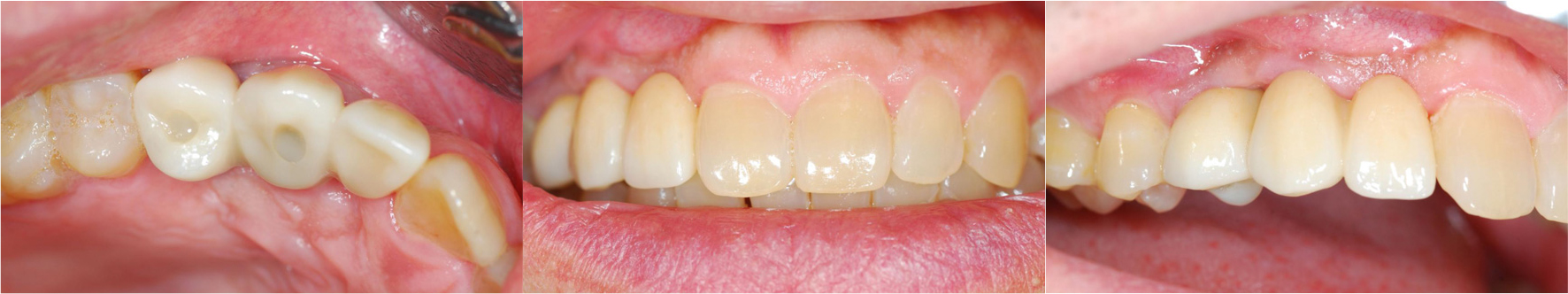
Prosthetic procedures were initiated after a 3-month osseointegration period. Customized abutments were fabricated and temporary restorations were delivered to verify occlusion and aesthetics. After confirming satisfactory function and patient acceptance, a final three-unit fixed dental prosthesis (#12 pontic, #13 and #14 implant-supported) was fabricated and cemented (Fig. 5).
The final restoration demonstrated a harmonious emergence profile with adequate soft tissue support, proper occlusal contacts, and satisfactory aesthetics that blended well with the adjacent natural teeth.
6. Follow-up
The patient was followed-up at regular intervals for three years postoperatively. At each visit, clinical and radiographic examinations were performed to assess the implant stability, peri-implant soft tissue health, marginal bone levels, prosthetic integrity, and patient satisfaction.
At the 3-year follow-up, the patient reported excellent function with no discomfort. Clinical examination revealed a healthy peri-implant mucosa with no signs of inflammation, a stable prosthesis with intact margins, and proper occlusal function.
Panoramic radiography demonstrated complete bone regeneration in the previously grafted area with excellent osseointegration of both implants (Fig. 6A). Marginal bone levels remained stable, with no significant bone loss. The grafted bone showed complete consolidation and maturation which appeared indistinguishable from native bone. A comparison of the initial and 3-year CBCT 3D reconstruction images further confirmed complete bone regeneration and maintenance of graft volume (Fig. 6B).
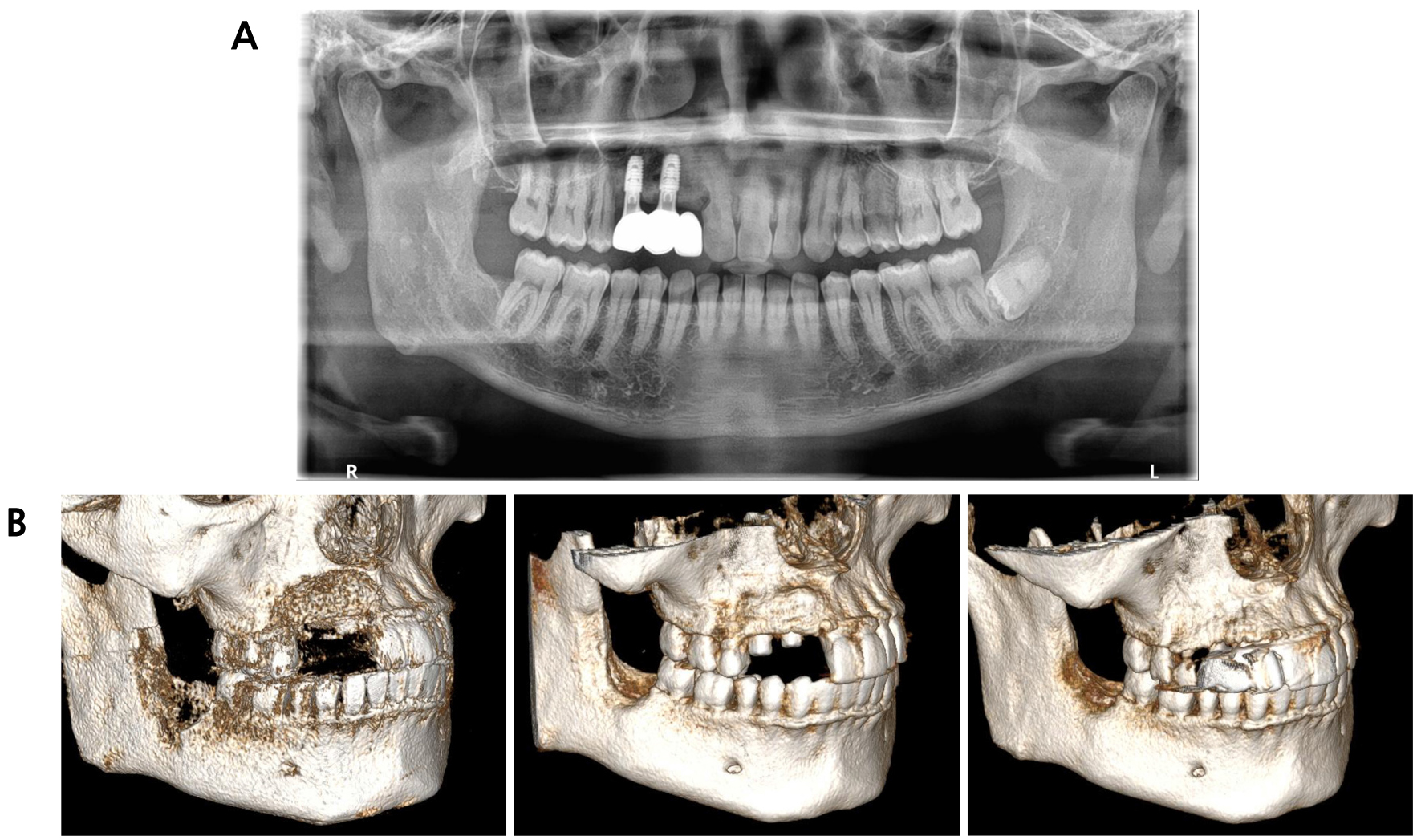
Fig. 6.
Three-year follow-up. (A) Panoramic radiograph demonstrating stable marginal bone levels, complete osseointegration, and maintenance of graft volume with no significant bone resorption, (B) CBCT three-dimensional reconstruction images immediately after initial bone graft (upper), after implant placement (center), and 3-year follow-up (lower).
Ⅲ. Discussion
This case report demonstrated the successful reconstruction of a maxillary segmental alveolar defect using ramal cortical bone combined with an allogeneic bone graft, followed by implant-supported rehabilitation. The 3-year follow-up confirmed complete bone regeneration, excellent implant stability, and maintenance of graft volume, highlighting several important clinical considerations.
Chemical burns from sulfuric acid can cause immediate tissue necrosis and delayed complications.9 In this case, the patient experienced a chemical burn 40 years before presentation; however, the resultant bone defect and root resorption only recently became clinically apparent. The defect likely formed during the acute phase of the chemical injury, subsequently healed with soft-tissue coverage, and remained stable for decades. The patient presented to the clinical after becoming aware of food impaction, and not the ongoing bone resorption. Notably, the defect showed no pathological lesions and was covered by healthy gingival epithelium indicating that the bone loss was a healed sequela of the original chemical injury, rather than an active infectious or neoplastic process.10
Segmental bony defects present unique reconstructive challenges owing to the loss of buccal and palatal cortical plates, which can compromise graft containment and stability.11 While various bone grafting materials have been developed, autogenous bones remain superior in terms of osteogenic potential, osteoinductivity, and osteoconduction.12 Ramal cortical bone offers distinct advantages including superior volume stability, intraoral harvesting with minimal morbidity, and inherent osteogenic potential.13,14 The cortical block provides structural support that resists resorption and maintains three-dimensional architecture, whereas viable osteoblasts within the graft contribute directly to new bone formation. In this case, combining a cortical block with particulate allogeneic bone created an optimal environment for predictable bone regeneration.
The use of the indigenous palatal soft tissue as a covering flap was critical to successful implantation of this case. A healthy, well-vascularized palatal mucosa provides a rich blood supply for graft integration, acts as a natural barrier membrane for graft containment, and enables tension-free primary closure.15 This approach of utilizing local soft tissue as a biological membrane may be superior to the use of synthetic membranes alone, offering both mechanical protection and biological enhancement of bone healing.
A combination of the ramal cortical block with particulate allogenic bone proved to be synergistic in this case. The cortical block provided immediate volume and structural integrity, whereas the particulate allograft filled the remaining defect space and enhanced the overall graft volume.16 This approach leverages the superior osteogenic properties of autogenous bone while minimizing donor-site morbidity by supplementing it with allografts.17
The 3-year follow-up demonstrated complete maintenance of the graft volume with no significant resorption, underscoring the value of cortical bone grafts in significant defects. Many studies have reported variable resorption rates with different graft materials, specifically particulate grafts and bone substitutes.18,19 The stability observed in this case suggested that the use of ramal cortical bone as a structural framework combined with healthy soft tissue coverage created optimal conditions for long-term graft stability.
Both implants achieved excellent primary stability at placement and demonstrated stable osseointegration throughout the follow-up period with no marginal bone loss or peri-implant complications. This outcome confirms that properly regenerated bone can support dental implants as effectively as the native bone.20
This case report has several important clinical implications. Chemical burn sequelae should be considered in the differential diagnosis of unexplained bone loss, even decades after the injury. Rammal cortical bone remains the gold standard for significant segmental bony defects requiring volume stability. At the same time, healthy indigenous palatal tissue can serve as an excellent biological covering that might eliminate the need for synthetic barrier membranes. With appropriate surgical techniques and material selection, complete bone regeneration can be achieved in complex defects, enabling successful long-term implant-supported rehabilitation.
In conclusion, this case demonstrated successful reconstruction of a maxillary segmental bony defect using the ramal cortical bone combined with an allogenic bone graft and indigenous palatal tissue as a biological covering. Complete bone regeneration and excellent volume stability were maintained for over three years, leading to successful implant-supported rehabilitation. This approach is a reliable and predictable treatment modality for complex alveolar defects in the aesthetic zone.