Ⅰ. Introduction
Extraction must be performed if necessary, depending on the tooth and periodontal condition, and appropriate restoration is performed according to the healing state of the extraction socket. In the healing of the tooth extraction site, epithelial formation is completed within 6 weeks,1 and bone formation proceeds for approximately 3−6 months.2 In the healing process of tooth extraction, if the epithelialization of tooth extraction is poor for various reasons, it can negatively affect bone formation. Therefore, a stable soft tissue covering the extraction socket is essential for subsequent implant-related procedures.
Hypertrophic scars, defined as thick raised scars, are abnormal wound-healing responses that exhibit stratum corneum barrier dysfunction and benign fibrous growths during wound healing.3 A hypertrophic scar shows a similar histological feature to that of keloid on light microscopy; however, it is known that biochemical and molecular differences exist.4 A hypertrophic scar is usually caused by trauma, inflammation, surgery, or burns,5and is known to occur in any part of the body, although it is most common in cross joints or skin creases at a right angle.6
Reports related to scar formation in the oral cavity are mainly on those that have occurred in the alveolar mucosa after apical surgery7 or that keloid-like healing may occur after root coverage.8 In addition, among the reports of various healing abnormalities in extraction sockets, to the best of the author’s knowledge, no reports of hypertrophic scarring exist.
In 2006, Adeyemo et al. reported healing of 318 extraction sockets, of which only one extraction socket had hypertrophic scar/keloid formation.9 Although hypertrophic scarring has a very low probability when it occurs, it will be a case in which the operator will be concerned about treatment. Therefore, the purpose of this case report is to share the treatment and healing progress of hypertrophic scars that occurred in the first and second molar extraction sockets.
Ⅱ. Case Report
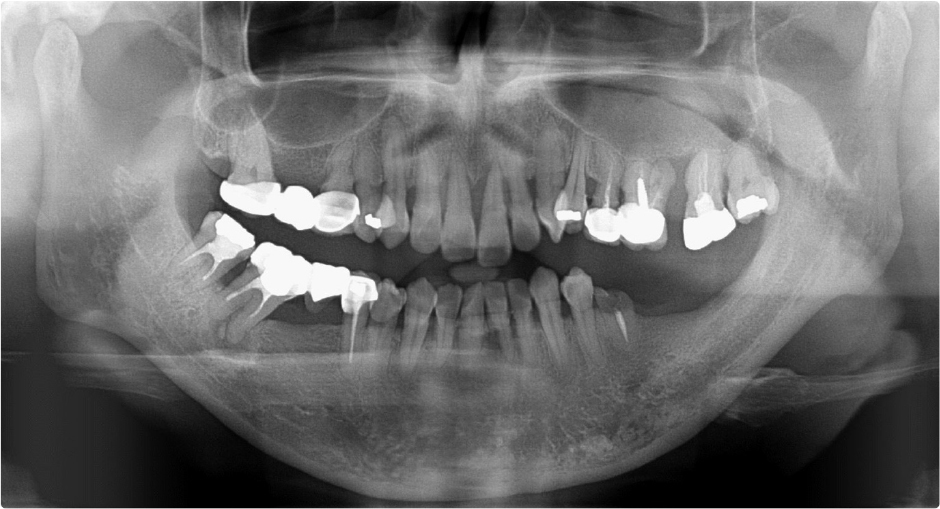
A 42-year-old female patient presented to our hospital with mobility of the maxillary incisors and molars. Clinical examination revealed grade 3 mobility in the maxillary left incisor and grade 2 mobility in the right incisor and left molars. Radiographic examination confirmed vertical and horizontal alveolar bone resorption around the maxillary incisors and horizontal alveolar bone resorption around the left molars (Fig. 1). The teeth were diagnosed as hopeless and were extracted for subsequent implant placement.
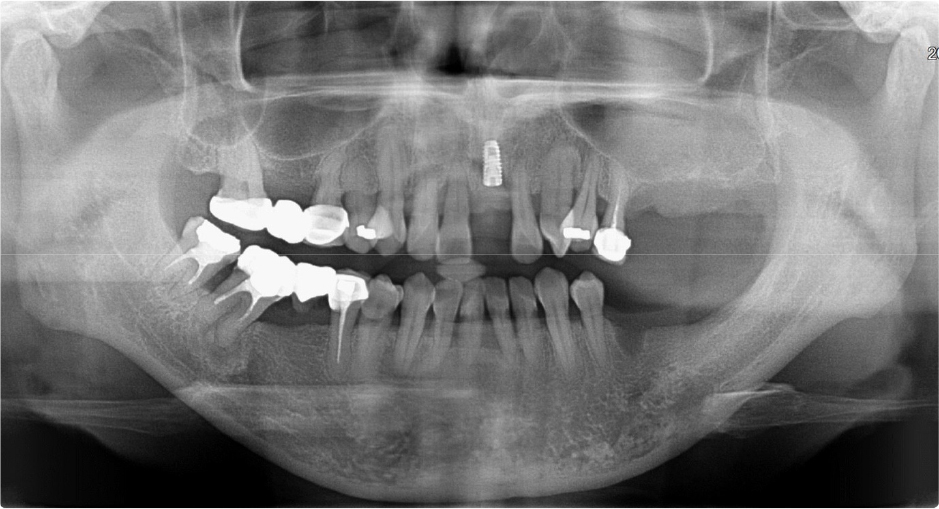
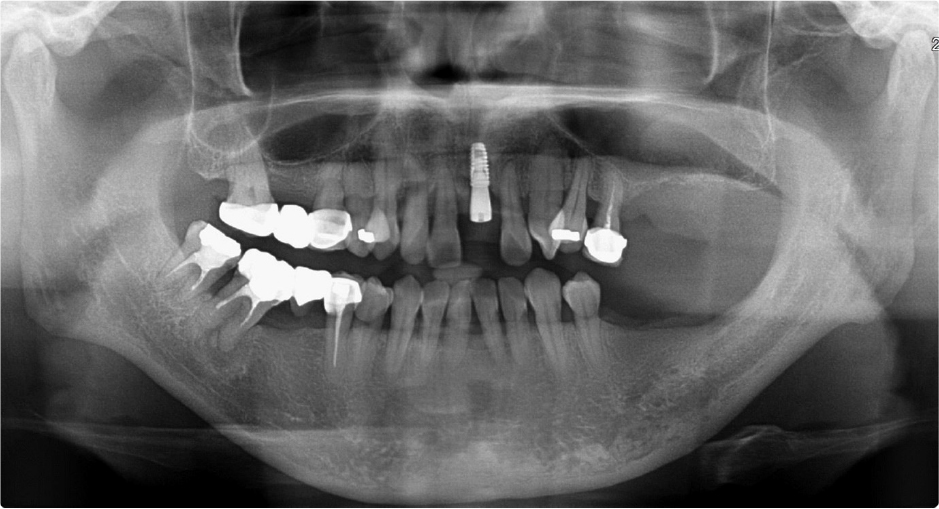
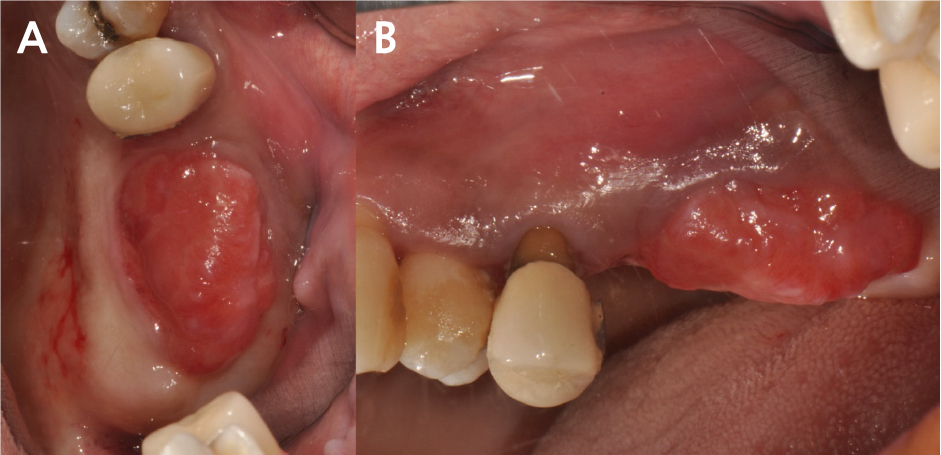
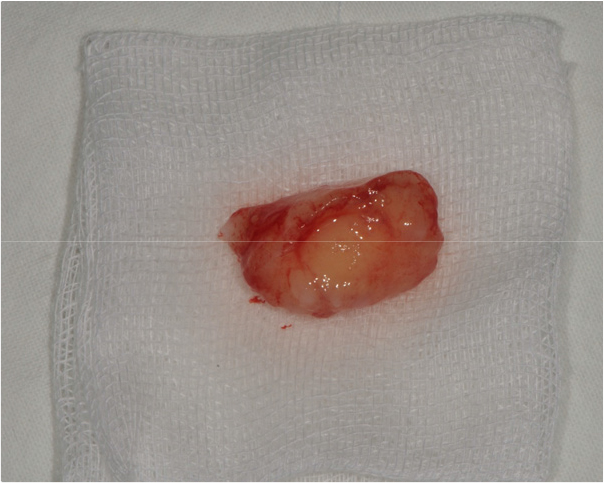
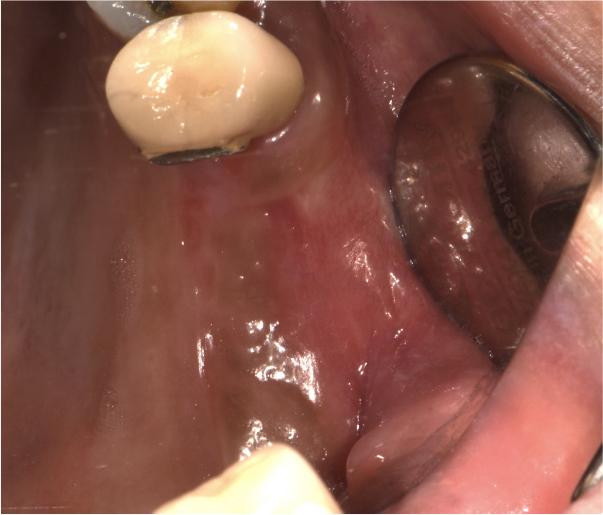
Two months after the extraction, slight soft tissue swelling was suspected on the panoramic view of the maxillary left molar area (Fig. 2). Six months after extraction, the soft tissue swelling at the extracted first and second molars area was enlarged enough to be observed on the panoramic radiograph (Fig. 3), and an oval-shaped nodule with a length of 2 cm, width of 1 cm, and height of 1 cm could be observed with the naked eye (Fig. 4). An excisional biopsy was planned for the soft tissue nodule.
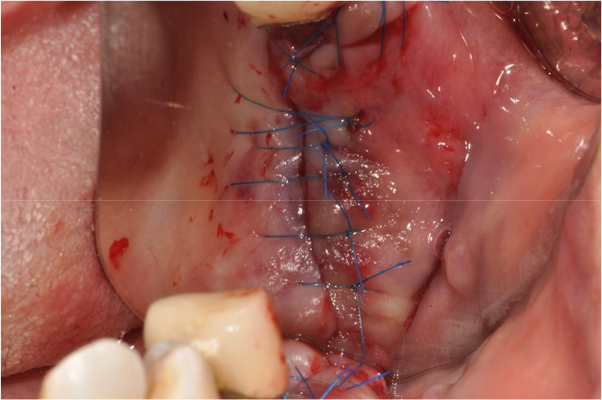
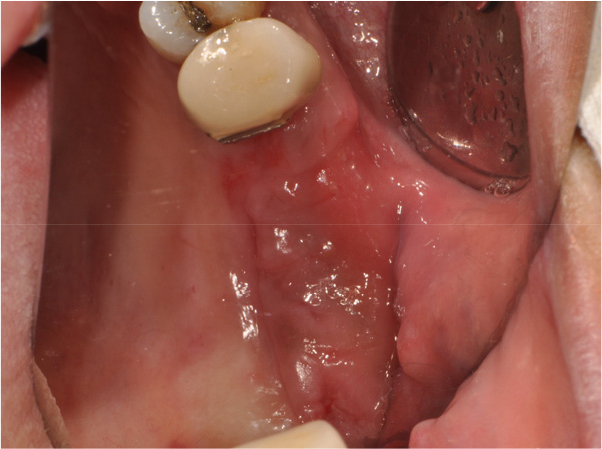
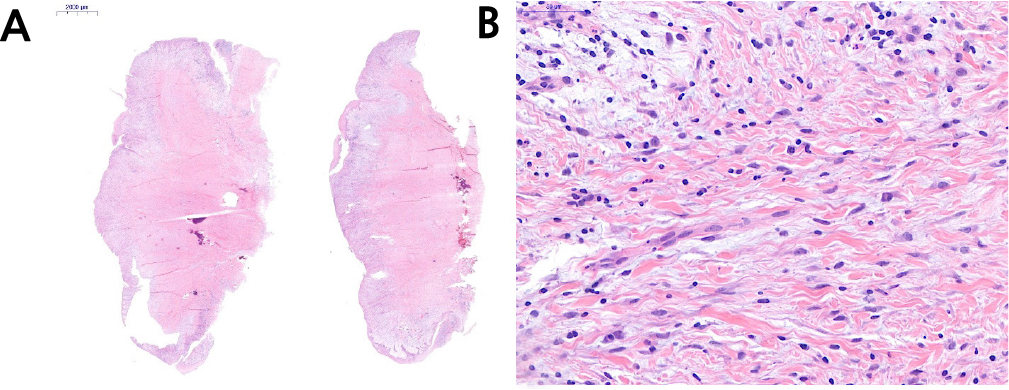
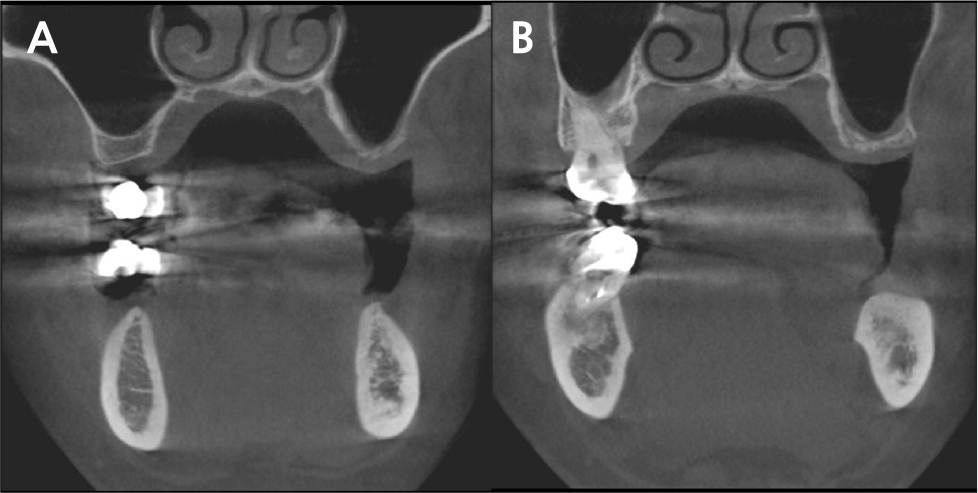
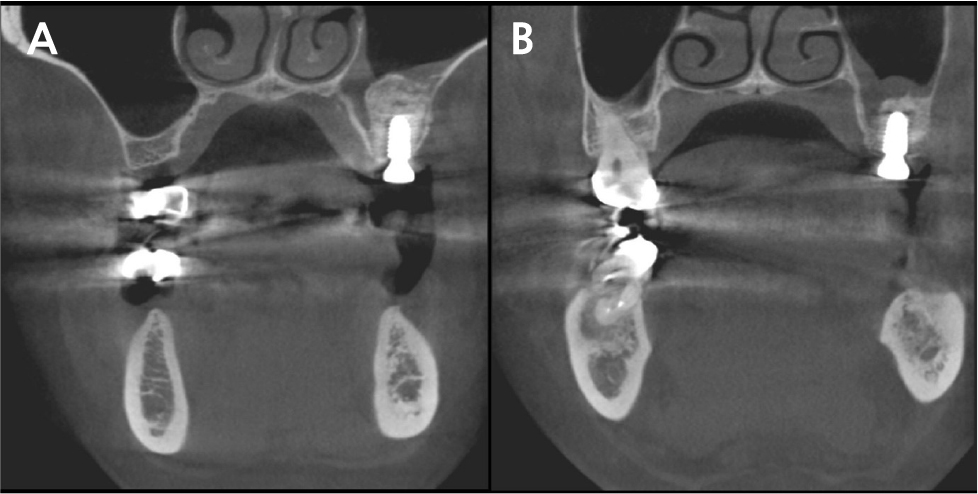
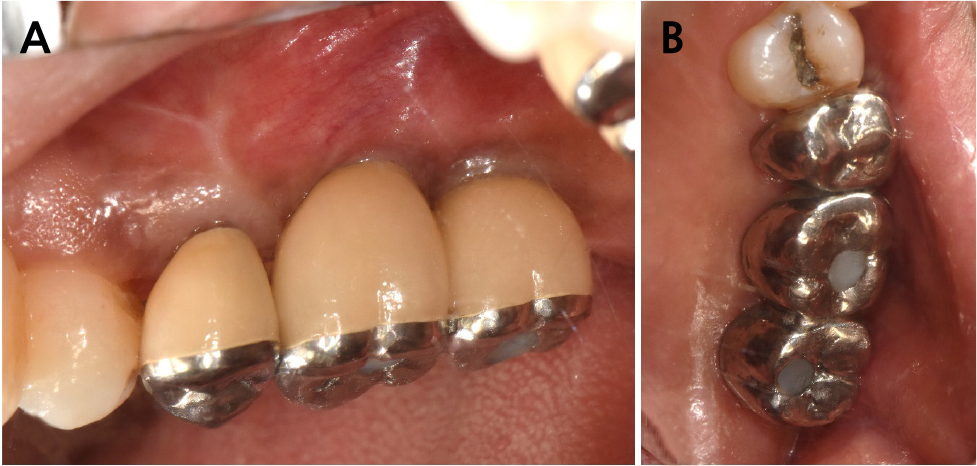
The patient was instructed to rinse with 0.12% chlorhexidine digluconate solution (Hexamedine; Bukwang, Seoul, Korea) for 2 min for oral disinfection. After local anesthesia with 2% lidocaine containing 1:80,000 epinephrine (Huons Lidocaine; Huons, Seoul, Korea), the nodule was excised, including approximately 1.5 mm of normal tissue with the periosteum (Fig. 5). A flap including a mesiobuccal vertical incision was elevated to achieve primary closure of the excised tissue. Subsequently, efforts were made to achieve primary closure of the flap as much as possible through simple interrupted sutures and modified Laurell suture with a 5-0 non-resorbable nylon monofilament (5-0 Ethilon; Ethicon, Cincinnati, OH, USA) (Fig. 6). At stitch removal after 2 weeks, healing was uneventful without any sign of inflammation (Fig. 7). Histological examination confirmed that it was a hypertrophic scar characterized by a nodule-type lesion and a wavy collagen bundle, with abundant keloidal collagen and eosinophil infiltration. (Fig. 8).6, 10 Two months after excisional biopsy, it was confirmed that there were no more soft tissue inflammatory or hypertrophic reactions (Fig. 9). Moreover, it was confirmed that there was no abnormality in the Schneiderian membrane of the maxillary sinus on cone-beam computed tomography (CBCT) (Fig. 10). Accordingly, sinus floor elevation with a lateral approach was performed, and there were no significant abnormalities even 6 months after implant placement (Fig. 11). Approximately 16 months after prosthesis placement, no specific abnormalities were observed when evaluated radiologically and clinically (Fig. 12).
Ⅲ. Discussion
Extraction is a procedure that must be performed depending on the tooth and periodontal conditions. The extraction socket is generally covered with soft tissue within 6 weeks; therefore, the internal bone formation pattern is usually evaluated radiologically. However, in this case, hypertrophic soft tissue was confirmed 6 months later. In addition, through a case review, it was confirmed that there was a suspicious pattern of soft tissue swelling that could not be visually confirmed, even in the panoramic view, 2 months after tooth extraction. However, as the bone in the lower margin of the maxillary sinus was confirmed on the radiograph 6 months after tooth extraction, it was determined that soft tissue proliferation did not interfere with bone formation or induce bone resorption.
The patient affirmed that keloid-like healing was observed several years ago during childbirth-related surgery. However, a study showed that pregnancy stimulates the keloid reaction;11 therefore, the patient’s general condition was determined to be different from when the patient was pregnant. Therefore, although extraction and implant surgery are possible, the author was informed about the possibility of adverse reactions. After tooth extraction, soft tissue healing of the extraction sockets showed keloid-like reactions. If the keloid were correct, it would be difficult to treat, and the risk of recurrence would have been relatively high, even if it was removed. However, as a result of checking several journals, the author learnt about hypertrophic scars that are similar to keloids, but have slightly different characteristics, and were clinically judged to be closer to this. The difference that can be easily identified clinically is that, unlike keloids, a hypertrophic scar does not show a pattern in which the proliferated tissue invades the surrounding normal tissue, and it exhibits a pink color without dark pigmentation, which is consistent with this case. Table 1 summarizes the clinically determinable differences between hypertrophic scars and keloids.4, 6, 12
Table 1.
Clinical differences between hypertrophic scars and keloids
There are various methods to treat hypertrophic scars, including occlusive dressings, compression therapy, excision, and cryosurgery. However, considering the treatment period for implant placement, special environment of the oral cavity, and method that can be performed by a dentist, removal of the hypertrophic scar through excisional biopsy was identified to be the most reasonable. In addition, surgery was performed to prevent the recurrence of hypertrophic scars by attempting to achieve as much primary wound closure as possible.12 Accordingly, it is considered that results without recurrence were observed.
The biopsy result showed that a hypertrophic scar was suspected; however, the possibility of odontogenic fibroma was also mentioned. The healing progress was reviewed over time, and the rest of the procedure was performed. When extraction was previously performed, soft tissue abnormalities were also observed in the panoramic view 2 months after extraction. After a healing period of approximately 2 months, it was confirmed that it was maintained without any abnormal findings, and subsequent procedures were performed. No hypertrophic scars were observed in the healing of the subsequent invasive procedures (maxillary sinus floor elevation with lateral approach and implant placement), and abnormal thickening of the maxillary sinus mucosa was not observed on CBCT. Currently, no specific adverse reactions have been observed 16 months since the completion of the prosthesis.