Ⅰ. Introduction
Midfacial defects, including those of the maxilla and palatine bones, frequently occur because of head and neck tumor resection. Midfacial defects are some of the most difficult surgical defects to manage not only in terms of disease control but also in achieving oral and facial reconstruction and rehabilitation.1
With the development of microsurgical techniques and experience, free-flap surgery using the scapula, fibula, forearm, anterolateral thigh, and rectus abdominis muscle as donor sites has been used for maxillary palatal defect reconstruction.2,3,4,5 Among these various free flaps, the scapular flap has several important advantages in the reconstruction of maxillary bone defects.
The scapular tip is morphologically similar to native maxillopalatal anatomic structures and can be applied to defects resulting from maxillectomy.6 Donor-site morbidity, including restricted shoulder movement, is limited compared to other reconstructive options.5,6,7 Several clinical studies have demonstrated the suitability of dental implants. A thin plate at the center of the scapula can be used for reconstruction of the hard palate, and the lateral border of the scapula is appropriate for alveolar bone reconstruction because its size and morphology allow sufficient dental implant placement.8,9,10
Placement of dental implants can dramatically improve the quality of life of patients with cancer. Patients who have been unable to use dentures and can only consume a soft or liquid diet can regain occlusion as close as possible to their natural teeth by undergoing fixed prosthetic treatment after a dental implant surgery.11,12
Here, we report a case of functional rehabilitation using a dental implant after reconstruction with a scapular free flap following the surgical removal of squamous cell carcinoma of the maxillary gingiva.
Ⅱ. Case Report
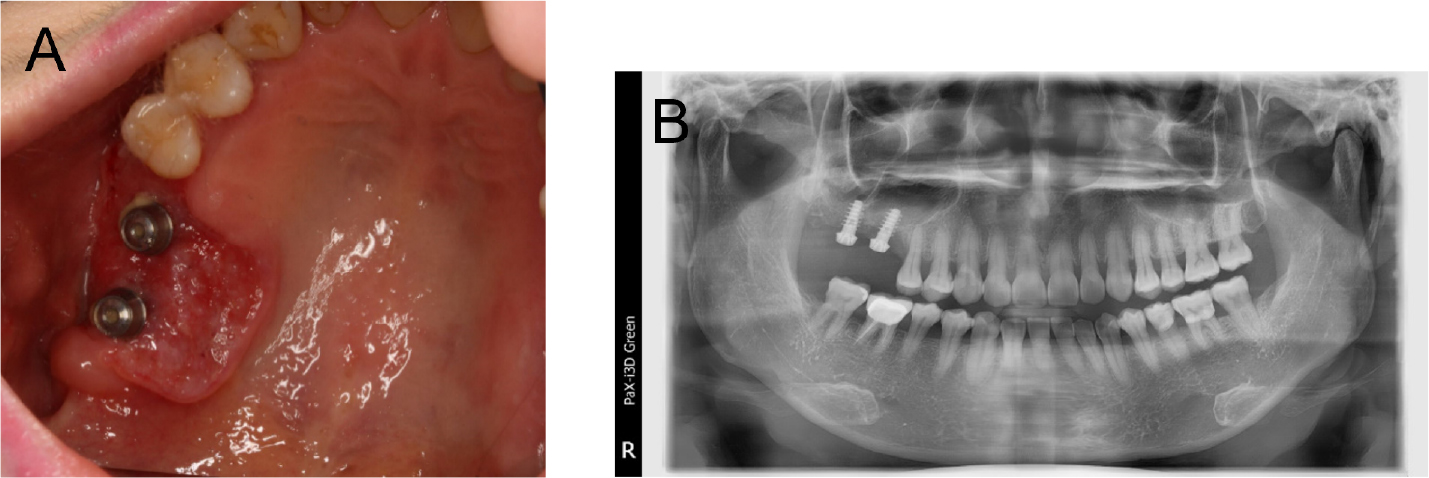
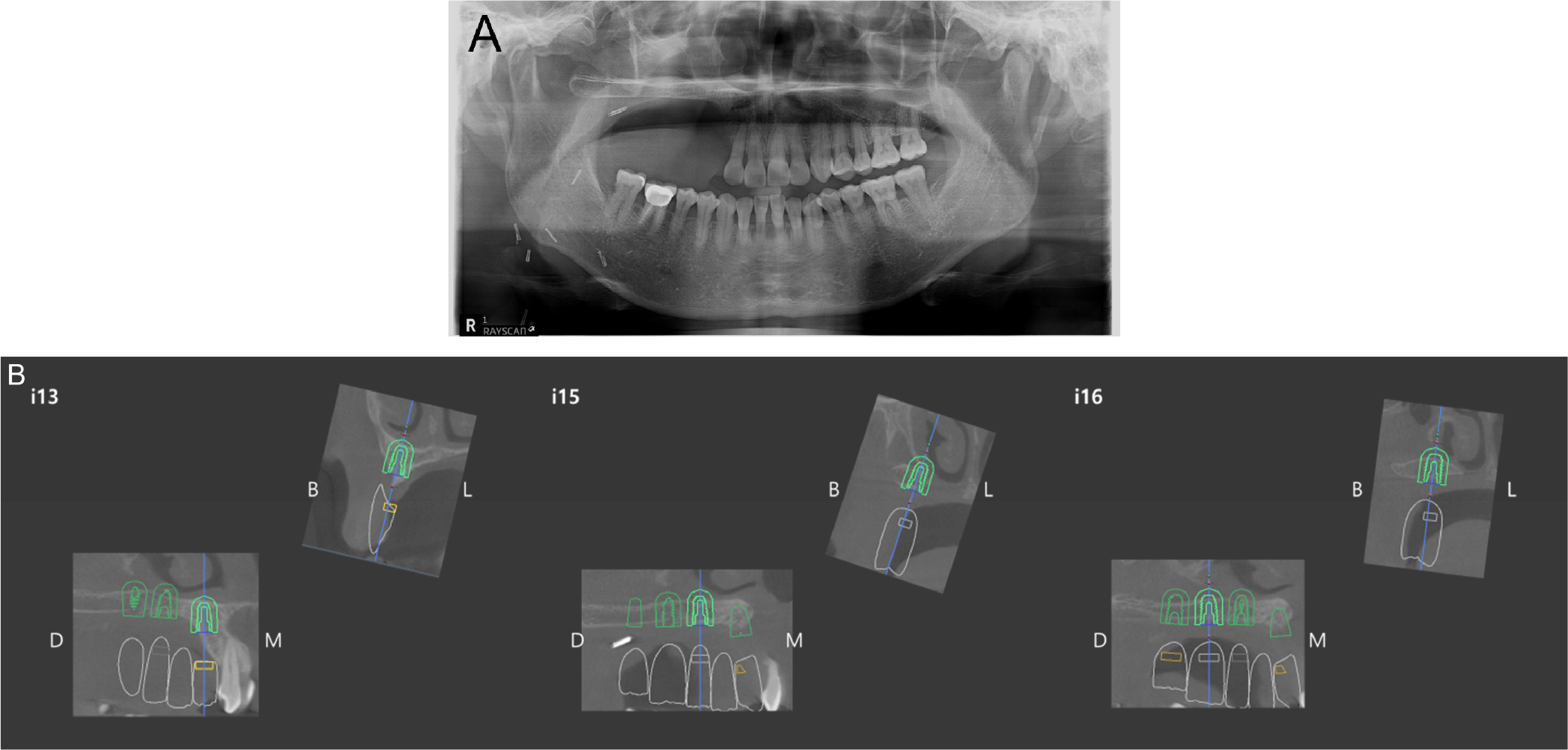
On December 10, 2019, a 56-year-old woman was referred to the Department of Oral and Maxillofacial Surgery at the Dental Hospital of Yonsei University with inflammatory symptoms in the right upper molar region. Examination revealed ulcerative lesions with indurations around previously placed dental implants (#16i and #17i). Discontinuity of the right maxillary sinus floor and alveolar bone loss around implant #16i and #17i were observed on the initial panoramic radiograph (Fig. 1). Incisional biopsy of the area revealed squamous cell carcinoma.
Computed tomography (CT), magnetic resonance imaging (MRI), and 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) were performed for further assessments. The MRI showed an infiltrative enhancing lesion in the right hard palate and gingival area. The PET/CT showed increased FDG uptake involving the right hard palate and adjacent gingiva. No abnormal increase in FDG uptake was observed, thereby suggesting lymph node or distant metastases (Fig. 2). Based on the above results, partial maxillectomy involving the right side of the maxilla and supraomohyoid neck dissection on the right side were planned. Additionally, reconstruction using a scapular free flap was planned to promote dental implant placement while minimizing donor-site morbidity.
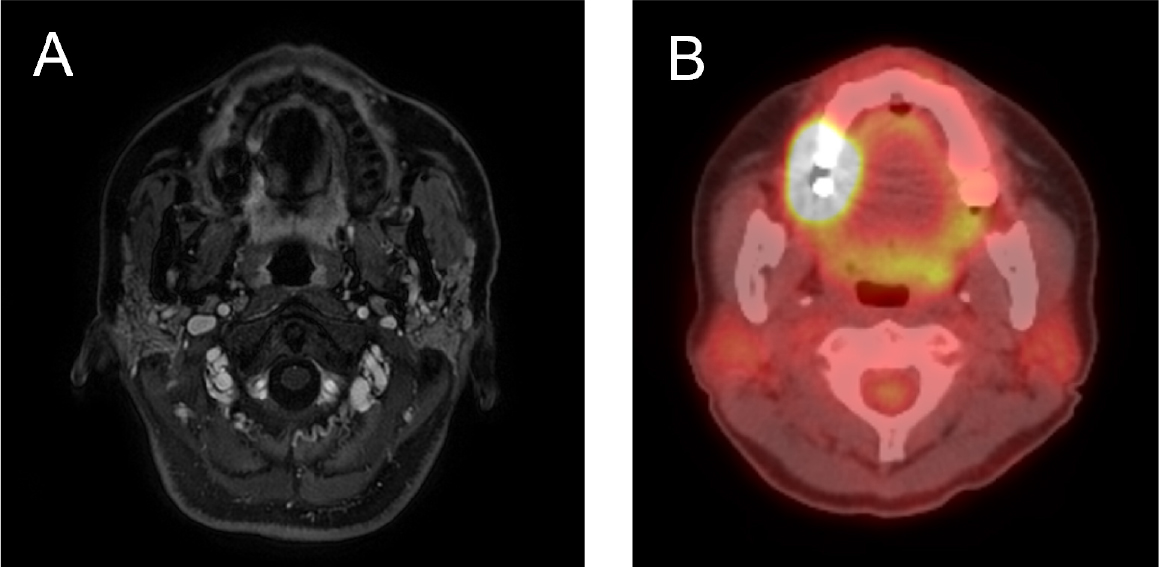
Fig. 2.
(A) Axial view of magnetic resonance image showing enhancing lesions in the right hard palate and gingival area, (B) Axial view of a PET-CT scan showing increased FDG uptake involving the right hard palate and adjacent gingiva. FDG, fluorodeoxyglucose; PET-CT, positron emission tomography- computed tomography.
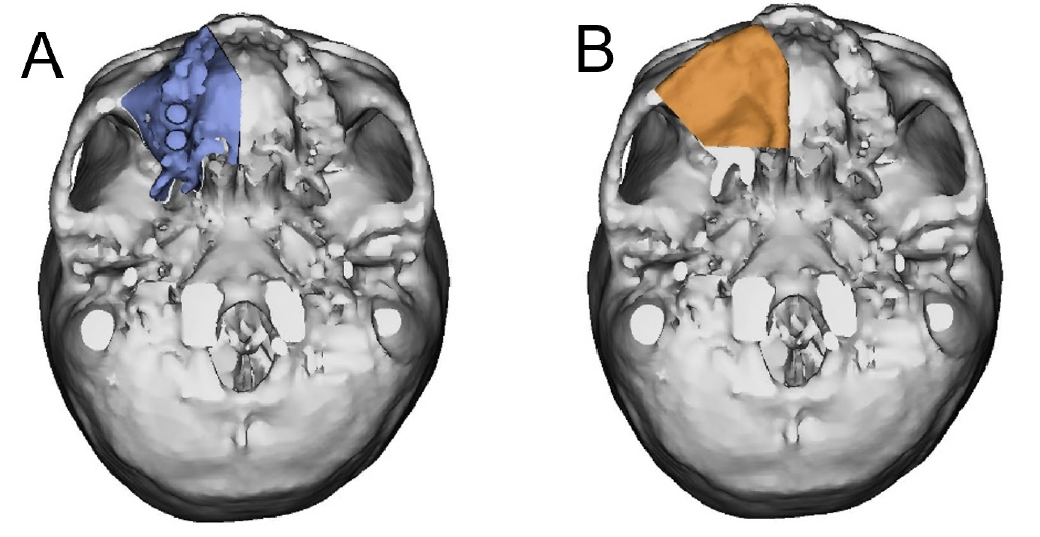
Before the reconstruction with the scapular free flap, virtual surgical planning (VSP) was performed using Mimics (version 16.0; Materialise Dental NV, Leuven, Belgium) and Simplant Pro 14.0 software program (Materialise Dental NV). The patients’ preoperative head and upper extremity CT scans were segmented, and the resulting three-dimensional models of the maxilla and scapula were loaded into the Mimics software for VSP. A partial maxillectomy was performed virtually, and the scapular model was positioned to replace the defect (Fig. 3). The height and width of the lateral border of the scapula were sufficient for the implant placement. Therefore, reconstruction of the lateral border of the scapula on the alveolar side was planned.
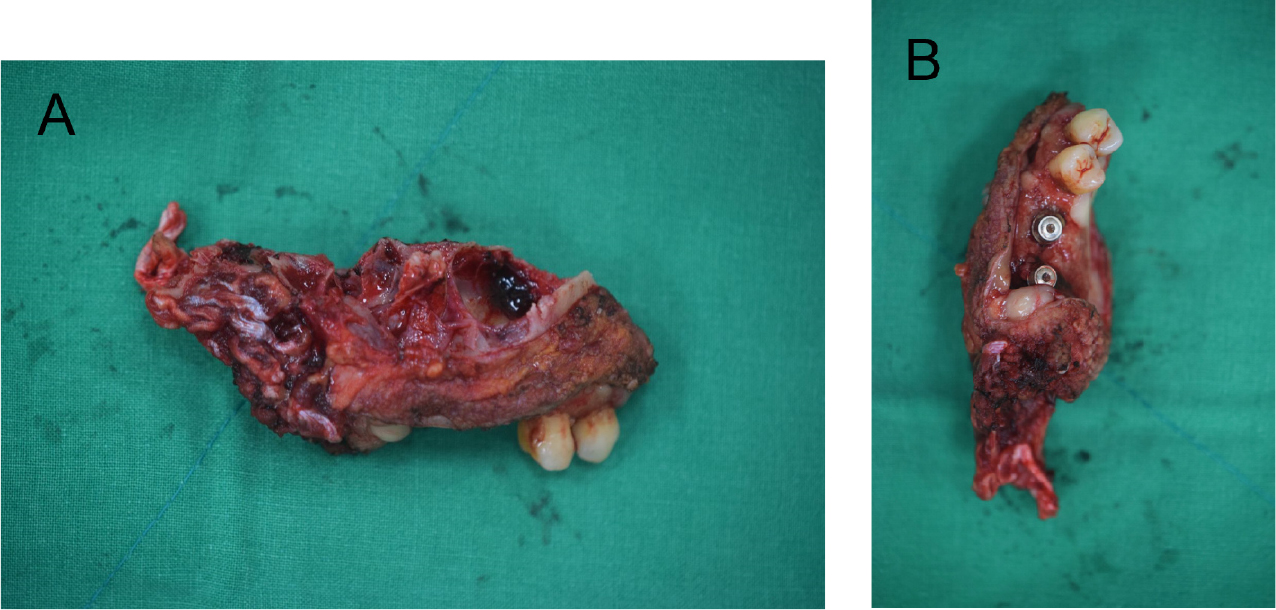
On December 30, 2019, a partial maxillectomy, supraomohyoid neck dissection, and reconstruction with a scapular tip free flap were performed under general anesthesia. To ensure a safety margin of 1 cm during the tumor resection, the upper right canine was extracted, and the extraction sockets of the upper right canine, pterygoid plate, and midpalate were determined as the resection margins (Fig. 4). After resecting the tumor, a frozen biopsy was performed, including that of the sinus mucosa, and all the tissues were reported to be tumor-free. Conventional supraomohyoid neck dissection was performed, and the scapular tip was harvested with the teres major muscle to reconstruct the oral mucosal defect. The patient was diagnosed with moderately differentiated squamous cell carcinoma, and the pathological stage was pT4aN0M0 stage IVA, according to the eighth edition of the American Joint Committee on Cancer (AJCC) staging system for head and neck cancers. After the radical surgery, the patient underwent postoperative radiotherapy for five weeks with an irradiation dose of 55 Gy.
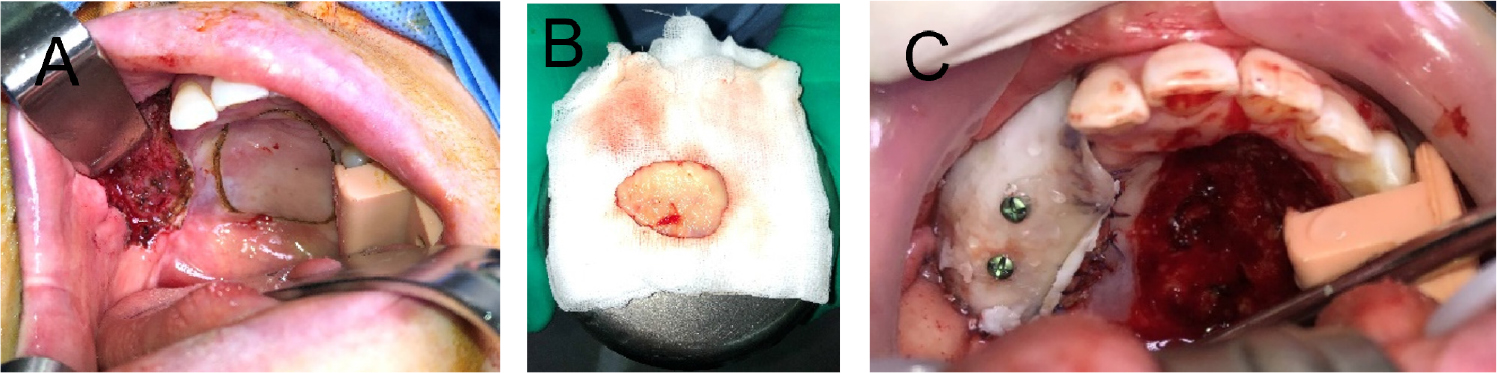
After hospital discharge, periodic clinical examinations were performed, and MRI performed every 3–6 months showed no recurrence of oral cavity cancer until 16 months postoperatively. On April 28, 2021, restoration of the right side of the maxilla with implants was discussed with the staff of the Department of Prosthodontics, and it was decided that the implants would be placed two years after the tumor resection. Due to postoperative scar contracture, vestibuloplasty was planned prior to the dental implant surgery. On July 8, 2021, vestibuloplasty and free gingival grafting were performed under general anesthesia, using the palatal mucosa as the donor site. Bone grafts, such as iliac bone grafts, were considered but not performed because the patient declined the procedure due to donor site complications. After scar identification, a blunt dissection was performed to form an appropriate oral vestibule, and the palatal mucosa was excised in the supraperiosteal plane to cover the dissected area. The palatal site was left granulated, and a prefabricated stent was affixed to the maxilla with screws to stabilize the wound and prevent relapse. The stent was left in place for one month, and was removed under local anesthesia (Fig. 5).
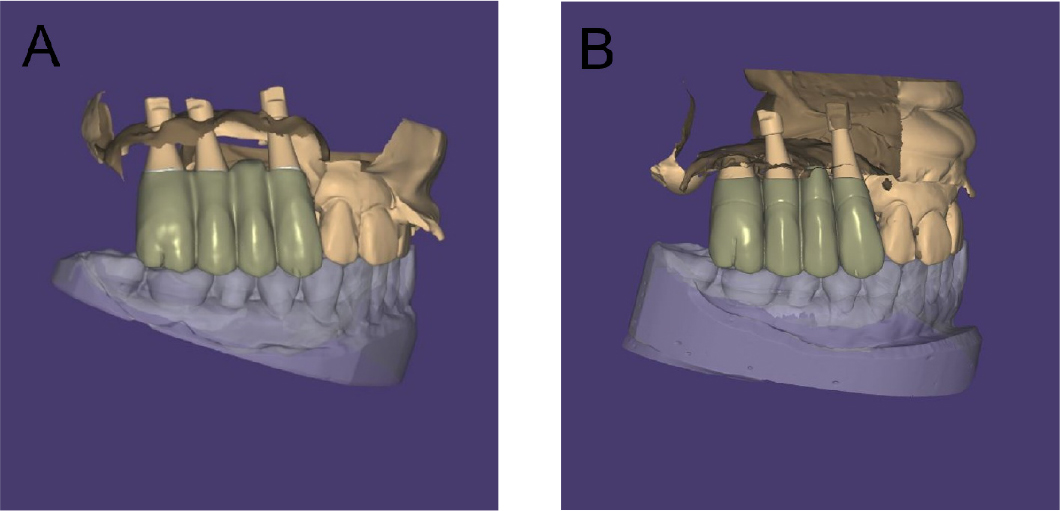
After confirming the absence of recurrence, a dental implant surgery was performed two years after the tumor resection surgery. The position of the implant was determined during consultation with the Department of Prosthetics, and a surgical stent was fabricated for the implant placement (Fig. 6). Three implants (#13i and #15i: IS III 4.5 × 8.5 mm, #16i: IS III 5.0 × 8.5 mm; Neobiotech, Seoul, Korea) were placed under local anesthesia. The bone width and height were adequately maintained during the implant placement. Among the three implants, #16i and #17i had excellent initial stability (>30 N); however, #13i did not show initial fixation. Therefore, a second-stage surgery was performed.
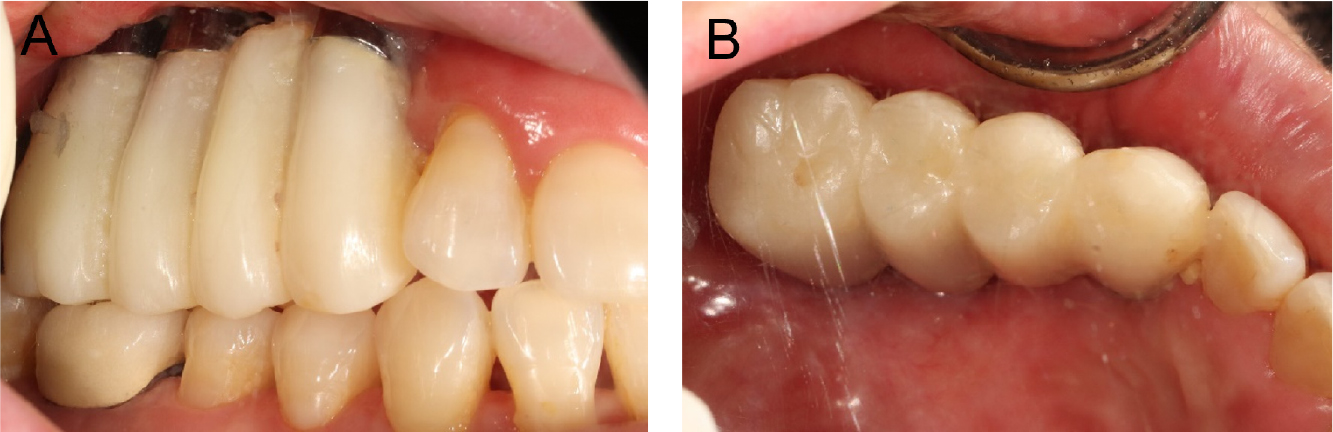
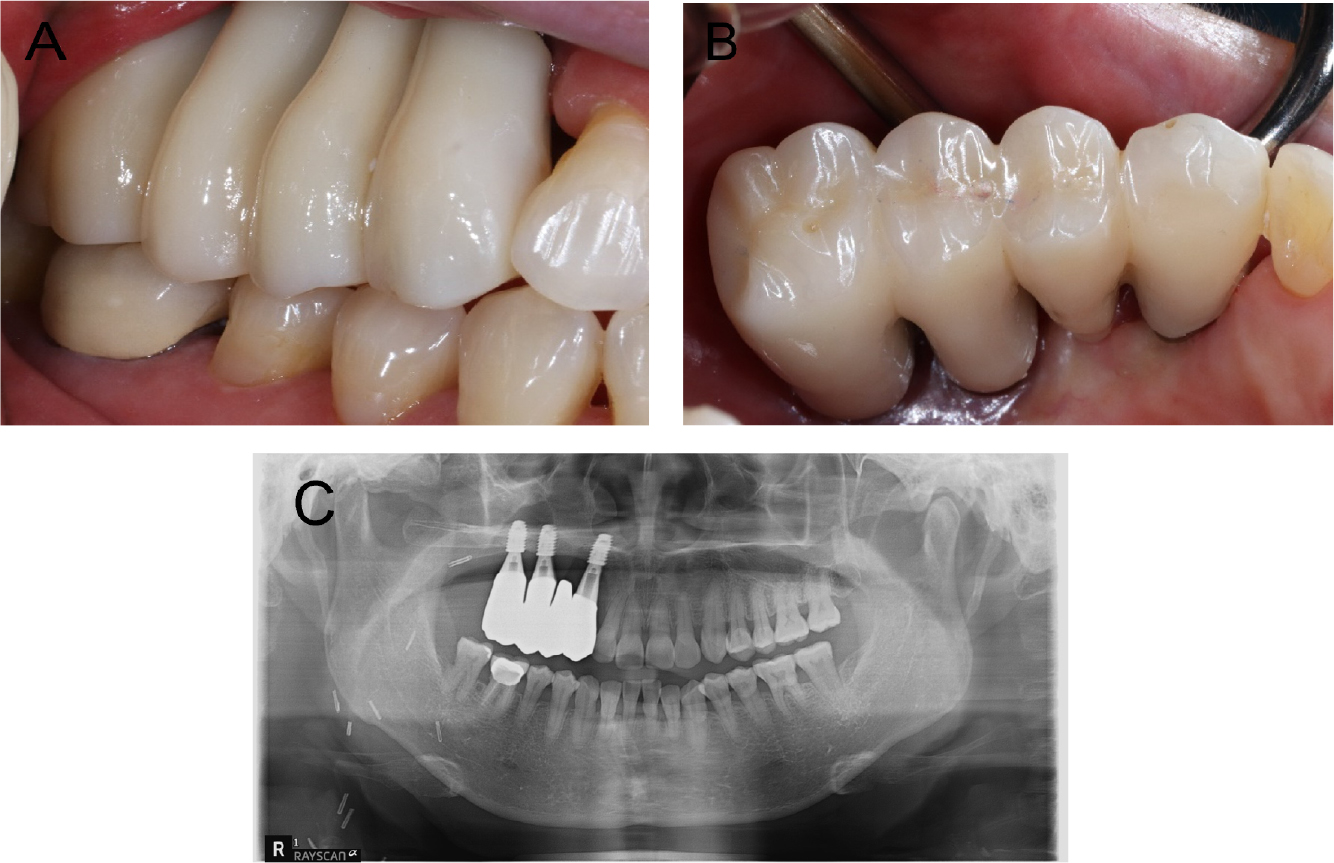
Conventional second-stage surgery was performed four months after the implant placement. Owing to the mucosal thickness, customized healing abutments were fabricated and connected to all the implant fixtures. Four months after the second-stage surgery, the implants were considered stable, and impressions of the provisional restoration were obtained. Impressions were made using an open-tray impression technique with pick-up impression copings and polyether impression materials (Impregum Penta; 3M ESPE, St. Paul, MN, USA). Five months after the second-stage surgery, customized abutments and a four-unit polymethyl methacrylate bridge were placed. During the provisional stage, the patients’ ability to adapt to the prosthesis and proficiency in maintaining proper oral hygiene were evaluated. After the prosthesis was placed, the patient reported difficulties in chewing on the right side and pronunciation problems. Over approximately four months, the patient gradually adapted to the provisional prosthesis. However, her oral hygiene management was poor, and persistent soft tissue inflammation was observed. Although toothbrushing instructions were provided at each visit, she had difficulty with cleaning the interproximal and pontic areas because of the long crown height. Therefore, to facilitate hygiene management, the customized abutment margin was positioned higher than the gingiva, and a new provisional prosthesis was fabricated with the prosthetic embrasure opened as wide as possible (Fig. 7). The patient used this new type of provisional prosthesis for approximately two months and visited the clinic for periodic check-ups. The patient adapted well to the new provisional prosthesis, and improvements were observed in her pronunciation, especially in affricate sounds and in her oral hygiene management skills. Additionally, she noticed an improvement in her chewing function, as she adapted to her new prosthesis. Once stable adaptation to the provisional restoration was achieved, a final prosthesis was fabricated, replicating the shape of the provisional prosthesis (Fig. 8).
Ⅲ. Discussion
In cases whereby maxillectomy is planned for patients with oral cancer, it is essential to consider the options of maxillary reconstruction. The importance of engaging prosthetic and surgical teams to provide patients with reconstructive options cannot be overemphasized.1 In this case, the patient required extensive resection and reconstruction for oral cancer of the upper gingiva. For future dental implant placement, vascularized free bone flaps, such as fibula, iliac bone, and scapular flaps, have been considered as treatment options. Among these options, the scapular free flap was selected for several reasons. First, the scapular free flap has negligible donor-site morbidity compared to other options. Although the teres major muscle was harvested together in this patient, there was no significant donor site morbidity, including limitation of arm movement or shoulder dysfunction postoperatively. Second, the anatomy of the scapula is similar to that of the hard palate and maxilla, making flap manipulation easy and practical.6 Additionally, because a dental implant can be placed in a scapular flap, it dramatically improves the restoration of occlusion and recovery of masticatory function after surgery.8,9,10 Therefore, dental implant location must be considered when reconstructing the mandible or maxilla with a scapular flap.
Therefore, virtual excisions and reconstructive surgeries are required. The VSP was performed using preoperative head and upper extremity CT scans. The VSP was used to confirm whether sufficient length and width for dental implant placement could be secured when the lateral border of the scapula was positioned as the alveolar ridge. The width of the lateral border of the scapula was 7–9 mm, which is sufficient for dental implant placement. Additionally, positioning of the scapular plate in the palatal defect was planned to ensure continuity of the bone defect. The actual surgery was performed in a similar manner as the virtual surgery, and the teres major muscle was harvested along with the flap to reconstruct the oral mucosal defect. The patient was diagnosed with moderately differentiated squamous cell carcinoma, and the pathological stage was pT4aN0M0 stage IVA, according to the eighth edition of the AJCC staging system for head and neck cancers. Radiation therapy was planned after surgery because the patient was diagnosed at an advanced stage. The patient underwent postoperative radiotherapy for five weeks at an irradiation dose of 55 Gy.
No recurrence was noted two years after the surgery; therefore, dental rehabilitation was considered. Since the patient underwent radiotherapy after surgery, there was a risk of osteoradionecrosis due to dental implant placement. There is no consensus in the literature regarding the threshold dose of radiation and the timing of dental implant placement, which may affect the survival of dental implants. However, previous studies have shown that the risk of osteoradionecrosis increases with doses that exceed 50 Gy.13 Furthermore, dental implant placement performed <12 months after radiotherapy may result in a higher risk of failure and osteoradionecrosis; however, no evidence from clinical trials has demonstrated this risk.14,15 Therefore, the implant placement was planned after providing a sufficient explanation of implant failure and obtaining informed consent from the patient.
In extensive oral defects repaired with free flaps, the soft tissue imported with the flap does not adequately replicate the inherent properties of a keratinized mucosa.16,17 Additionally, the patient had a narrow alveolar ridge due to tissue shrinkage and scar contracture. Therefore, soft-tissue management, vestibuloplasty, and free gingival grafting using the palatal mucosa as the donor site were planned and performed before the dental implant placement.
Dental implants were placed two years after the oral cancer surgery. Prior to the dental implant surgery, dental cone-beam CT was performed to evaluate the remaining bone status and fabricate the surgical stent, which revealed that the height of the remaining bone was 6–8 mm. Dental implants (8.5 mm height) were placed with a prefabricated surgical stent using bicortical anchorage to ensure a greater amount of bone-implant interface.
Masticatory function is not simply performed by the teeth, but by a combination of organized groups of structures, including the lips, teeth, soft tissues, and muscles.18 The patient in this case may have developed a long-term unilateral chewing habit because of a defect caused by the maxillectomy, resulting in decreased masticatory function. In such patients, even if the implant is stable, it is preferable to undergo a provisional prosthesis phase rather than proceeding directly to the fabrication of the final prosthesis. By incorporating a provisional phase, the prosthesis can be easily modified based on patient adaptability and oral hygiene management. In this patient, masticatory function gradually improved over approximately four months of provisional prosthesis use. However, there are limitations in oral hygiene management and persistent gingival inflammation. Excessive crown height can result in inappropriate oral hygiene management and chronic inflammation.19 Therefore, the provisional prosthesis was modified to facilitate better hygiene management, leading to improved patient adaptation to the implanted prosthesis. After the delivery of provisional restorations, regular clinic visits and oral hygiene education sessions were conducted. After confirming that the patient adapted well to the provisional prosthesis, the final prosthesis was placed (Fig. 9). Thus, implant prostheses in patients undergoing reconstructive surgery can be a more predictive treatment when provisional prostheses are used, and periodic examinations and evaluations are particularly important.
Ⅳ. Conclusion
Although this case report has limitations, including a small sample size and the possibility of overinterpretation, the authors reported that dental implants were successfully placed in patients who underwent ablative oral cancer surgery, scapular free flap reconstruction, and postoperative radiotherapy. After adapting to the provisional prosthesis, the patient underwent a definitive prosthesis installment and is currently satisfied with the functional and esthetic outcomes without any major complications.