I.Introduction
II. Materials and Methods
1. Animals
2. Fabrication of electrospun membranes
3. Analysis by scanning electron microscopy
4. Surgical procedures
5. Histological evaluation
6. Statistical Analysis
III. Results
IV. Discussion
V. Conclusion
I.Introduction
Dental implant therapy is known to be a successful treatment modality with a high predictability for partially or completely edentulous patients. However, adequate bone volume around the implant is one of the prerequisites leading to successful implants to provide primary stability and resistance to functional loading after the osseointegration1. A lack of horizontal and/or vertical bone in implant sites may result from infection, trauma, tumor resection, other deformities and tooth loss2. Large bone defects are often encountered when extraction has been performed due to severe periodontal disease or endodontic failure. All these situations can require alveolar ridge augmentation prior to or at the time of implant placement.
In most incidences, barrier membranes have been utilized in guided bone regeneration (GBR) procedure to maximize bone formation for the management of these bone deficiencies3. Barrier membranes are used to exclude rapidly proliferating epithelial cells and connective tissue cells inhibiting new bone formation and to allow the migration of osteogenic cells.
In general, barrier membranes are classified into two types according to their resorbability4. Lots of clinicians are in favor of resorbable membranes such as collagen membranes and synthetic polymers because they do not require additional surgery for removal5,6. Synthetic polyester-based membranes including poly (lactic acid) (PLA), poly (ε-caprolactone) (PCL) and poly (lactic-co- glycolic acid) (PLGA) have been actively researched because collagen membranes had some drawbacks such as uncertainty of the origin, relatively low mechanical properties and high price7,8.
As these synthetic polyester-based membranes could not have porous surfaces which is necessary for bone regeneration and angiogenesis, electrospinning method has been vigorously studied. Electrospinning could generate the structures similar to extracellular matrix7, having high porosity, controllability of mechanical and biodegradable properties9 and possibility of delivering genes or drugs10.
At this time, it is necessary to develop high-performance membranes on bone regeneration. Resorbable hydroxyapatite (HA)-coated micro/nanofibrous membranes fabricated from PCL and bioactive CaO-SiO2 gel solution by simultaneous electrospinning were prepared in the present study. It was hypothesized that slowly resorbable PCL microfibrous structures would maintain the space as a strong membrane scaffold and HA coating obtained from rapidly degradable bioactive nanofibrous CaO-SiO2 would give the membrane osteoconductivity which PCL did not have.
The aim of the present study was to evaluate the effects on bone regeneration of the resorbable HA-coated micro/nanofibrous membranes fabricated from PCL and bioactive CaO-SiO2 by electro-spinning in comparison with commercially available non-resorbable membranes and resorbable collagen membranes through the experiments of rabbit calvarial defects. Furthermore it was to assess the practicality of these HA-coated PCL/ CaO-SiO2 membranes and to apply for GBR procedures in the future.
II. Materials and Methods
1. Animals
Guided bone regeneration (GBR) procedures using three different barrier membranes were conducted on twenty two healthy New Zealand White male rabbits weighing between 2.0 and 2.5 kg. Throughout the experiment, the animals were individually housed in cages, fed ad libitum with a standard diet, and exposed to natural light/dark cycle. Rabbits were divided into three groups according to the type of used membranes. Non-resorbable membranes (Gore-tex®, WL Gore & Associates, USA; Group NR, n=8), resorbable collagen membranes (Ossix®, OraPharma, USA; Group RC, n=8) and HA-coated poly (ε-caprolactone) (PCL)/ CaO-SiO2 micro/nanofibrous membranes (Group HA, n=6) were used on calvarial bone defects of the rabbits. The protocol for animal experiments was approved by and performed in accordance with guidelines of the Institutional Animal Care and Use Committee of Seoul National University.
2. Fabrication of electrospun membranes
Resorbable HA-coated micro/nanofibrous membranes were fabricated from mixing microsized biodegradable PCL fibers with nanosized bioactive CaO-SiO2 gel fibers using electrospinning method.
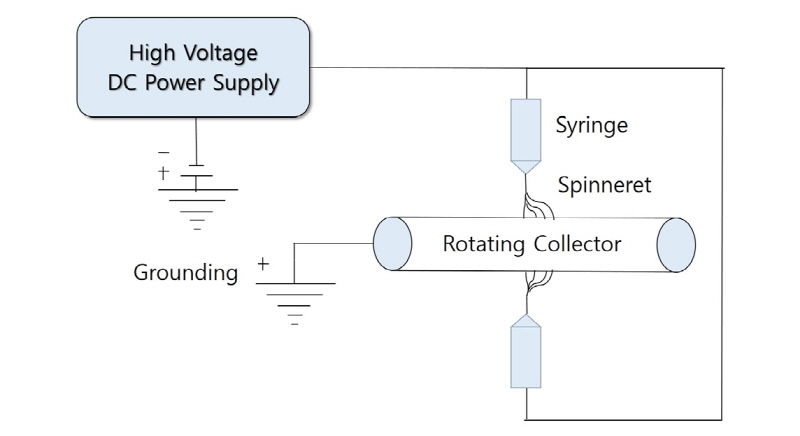
The CaO-SiO2 gel solution was prepared by consecutive condensation reaction at 80°C for 20 hours and 60°C for 2 hours and hydrolysis of tetraethyl orthosilicate (TEOS, Aldrich), the silica precursor with water, ethanol, calcium chloride dihydrate (Aldrich), and hydrochloric acid. By dissolution of PCL (Mw 80,000, Aldrich) in 1,1,3,3-hexafluoro-2-propanol (HFIP, Acros), 17% (by volume) PCL solution was prepared. The PCL and CaO-SiO2 gel solutions were placed to two different spinnerets tipped with 16-gauge needles and then a high electric field was generated. DC voltages of 10 and 20 kV were applied and the distances between each spinneret and the rotating collector were set by 10 cm for both of them (Fig. 1).
After the electrospinning, two mixed composite fibrous structures were dried for 24 hours, cut, sterilized under UV and then soaked in 30 mL of simulated body fluid prepared as indicated by Rhee et al.11 at 36.5°C for 7 days to allow hydroxyapatite coated on them. Finally, the fabricated HA-coated micro/nanofibrous membranes were cautiously rinsed with distilled water and dried.
In addition, commercially available two membranes corresponding to non-resorbable membranes (Gore-tex®, WL Gore & Associates, USA) and resorbable collagen membranes (Ossix®, OraPharma, USA) were prepared separately and the same procedure from soaking in simulated body fluid to rinsing and drying were performed.
3. Analysis by scanning electron microscopy
The surface morphology of the fabricated membranes was investigated by scanning electron microscopy (SEM) to identify whether the hydroxyapatites were properly coated. Two types of commercially available membranes were also analyzed for comparison.
4. Surgical procedures
Surgery was performed under both general and local anesthesia. First, general anesthesia of the rabbits was induced by an intramuscular injection of a 1:4 mixture of 3.5 mg/kg Xylazine (RompunⓇ, Bayer, Korea) and 7.5 mg/kg Ketamine (KetalarⓇ, Yuhan, Korea). After the calvarial skin was shaved and cleaned with a mixture of iodine and 5% chlorhexidine digluconate, local anaesthesia was induced by a subcutaneous injection of 2% lidocaine containing 1:100,000 epinephrine. About 4 cm longitudinal midline incision was made along the sagittal suture from the frontal area all the way to the occipital bone and skin-periosteal flap was reflected then. Using a trephine bur with 9 mm-outer diameter (3i-Implant Innovations, USA), the outline of the defect-to-be was definitely demarcated and the upper portion of bicortical bone within the demarcation was removed using rotary round bur with copious saline irrigation. Therefore, the thin cortical bone on the bottom of the calvarial defect was left to provide cell resources during bone healing process. Three types of membranes were placed over the created calvarial defect according to the corresponding group. Suturing was done layer by layer with bioabsorbable suturing materials for repositioning the periosteum and skin wound was closed with silk. After the surgical procedure, antibiotic injections were given for three days to prevent possible infection.
5. Histological evaluation
Histological and histomorphometric analysis were performed to evaluate the effect on bone regeneration and to assess the osteoconductivity of three types of membranes in vivo. Animals were sacrificed at 2 and 4 weeks after the surgery. The calvarial bones were fixed in a neutral buffered formalin solution, rinsed, dehydrated, and embedded in a media (Technovit 7200, Exakt Apparatebau, Germany) without decalcification. Then, the specimens were microsected, polished to 40µm thickness and stained with Polysciences Multiple Stain (Polysciences, USA). Histological features among three groups were compared under an optical microscope (Olympus, Japan) and histomorphometric measurements of newly formed bone were carried out using computer-assisted image analysis software (Tomoro Scope Eye 3.6, Techsan, Korea) based on histological features.
6. Statistical Analysis
Statistical analyses were performed by Kruskal Wallis test for intergroup analysis and Mann- Whitney U test for intragroup analysis. Analysis of Variance (ANOVA) testing was performed for additional reference. Data are presented as the mean±standard deviation (SD). P<0.05 was considered statistically significant.
III. Results
Hydroxyapatites coating was confirmed on the membranes made of PCL and CaO-SiO2 gel fibers, while it was not identified on the other membranes (Fig. 2A-2C).
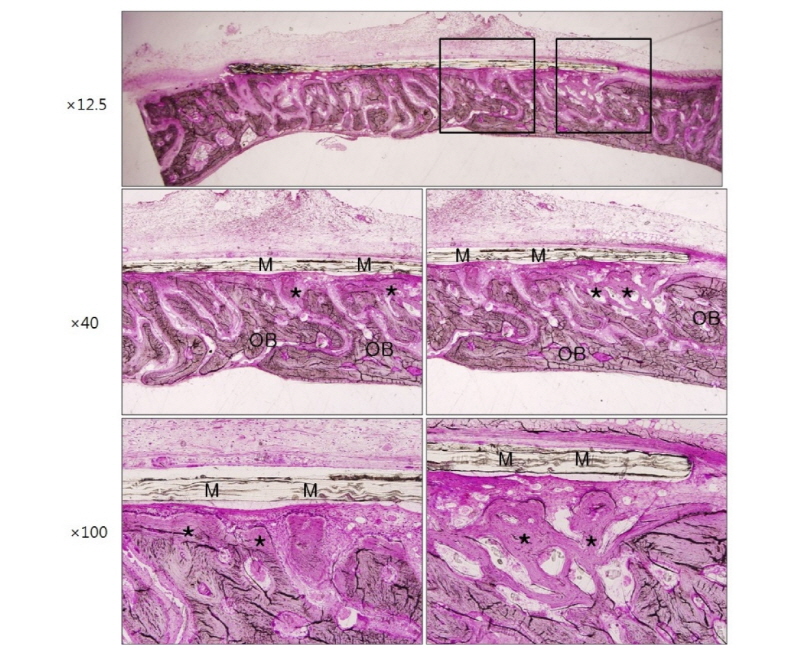
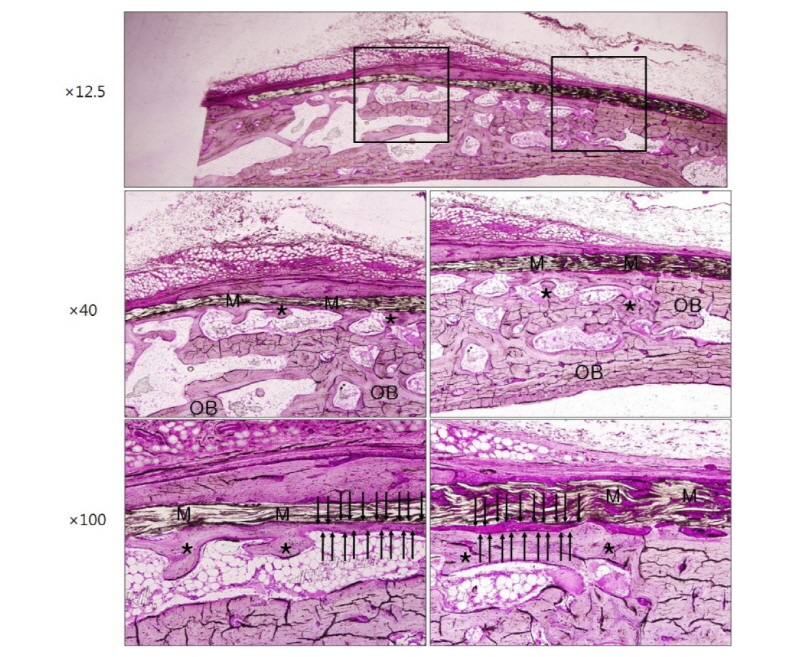
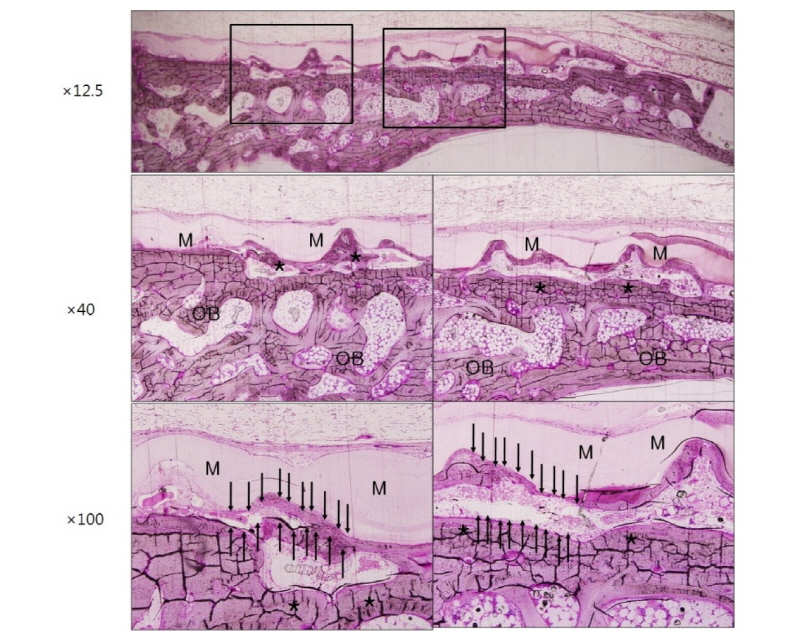
Healing was uneventful after the surgery and there was no evidence of infection in all three groups. Optical microscopic images corresponding to 2 weeks and 4 weeks after surgery in group NR using non-resorbable membranes are shown in figure 3 and figure 4, respectively. Each figure depicts the whole defect image, 40 times and 100 times original magnification images in order, from top to bottom (Polysciences Multiple Stain) (Fig. 3-8). Membrane, old bone and newly formed bone are indicated M, OB and an asterisk, respectively (Fig. 3-8). In specimen of group NR, it is shown that there are numerous new bone matrices and newly formed bone throughout all areas of the defect indicating robust bone regeneration at 2 weeks after surgery (Fig. 3, asterisk). The specimen of 4 weeks after surgery in the same group shows that most of the defects are filled with new bone over the old bone and maturation of new bone is in progress (Fig. 4, asterisk). Here, a noteworthy feature is that some fibrous tissue intervened between the membrane and bone tissue (Fig. 4, black arrows), which indicates that the membrane has no osteoconductivity in group NR.
Figure 5 and figure 6 show microscopic images at 2 weeks and 4 weeks after surgery in group RC using resorbable collagen membranes. In this group also, it is recognized that new bone formation is active over the entire area of the defect. It is shown that the shape of resorbable collagen membrane is fully maintained until 4 weeks after surgery (Fig. 6) and there is also the fibrous tissue intervention (Fig. 6, black arrows) as in group NR.
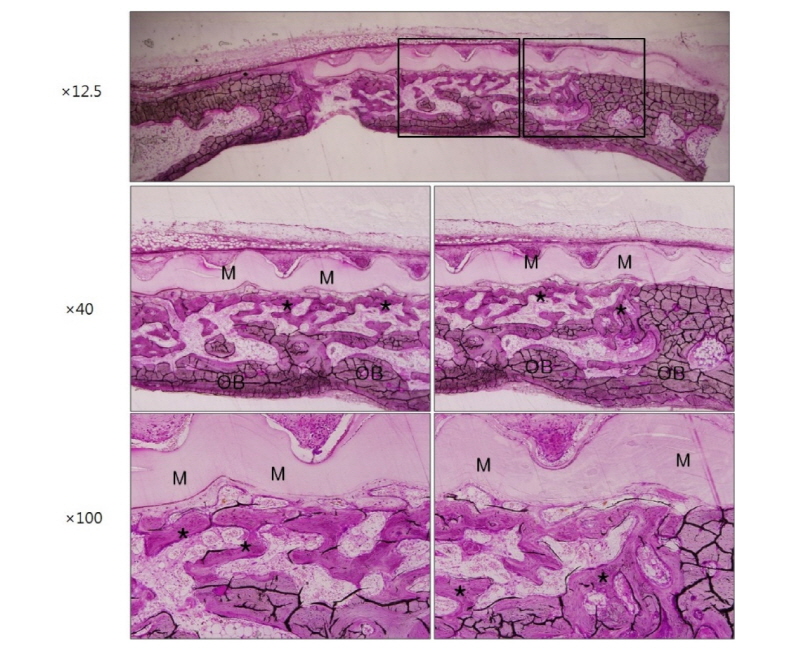
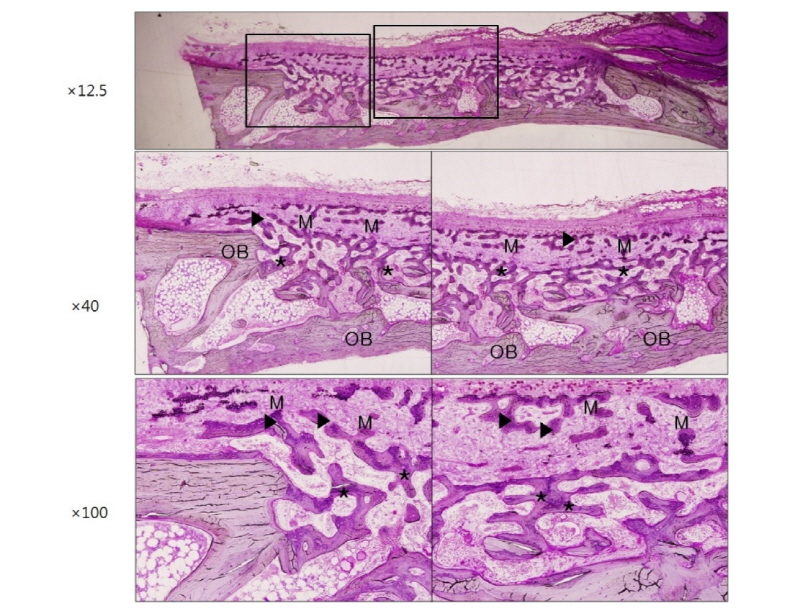
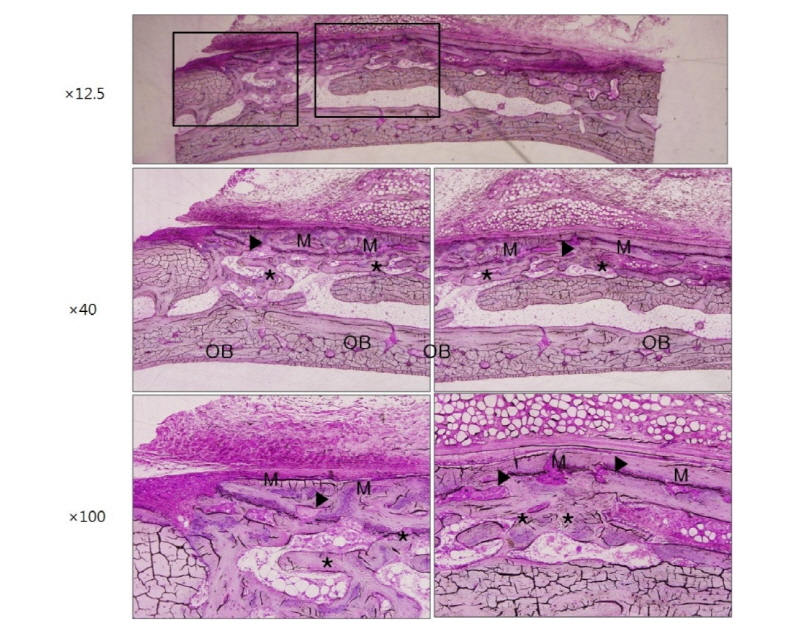
Finally, microscopic images are shown in figure 7 and figure 8 indicating the histological features of 2 weeks and 4 weeks after surgery in group HA. These images also show that large amount of newly formed bone exists all over the defect area like other groups. Ongoing maturation of new bone is well recognized on images of 2 and 4 weeks after surgery (Fig. 7, 8, asterisk and black arrowheads). In group HA, a remarkable feature was that new bone formation was under way directly beneath the membrane or as closely incorporated pattern into the membrane structure indicating that the membrane has high osteoconductivity and remarkable promoting effect on bone tissue integration in vivo (Fig. 7, 8, black arrowheads).
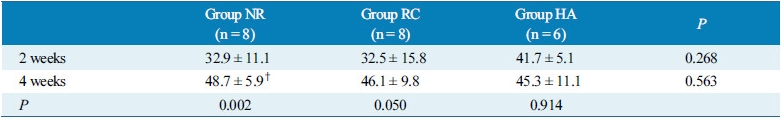
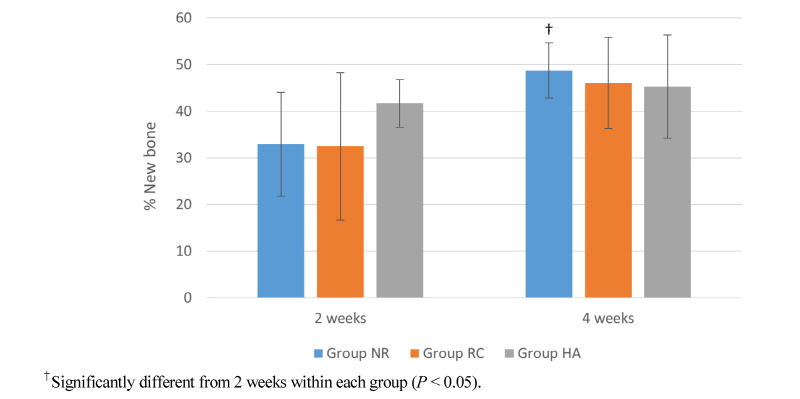
Table 1 and figure 9 show the results of histomorphometric analysis performed at 2 and 4 weeks after surgery. In group NR, the percentage of newly formed bone in the total defect area was about 32.9±11.1% and 48.7±5.9% at 2 and 4 weeks, respectively. These were 32.5±15.8% and 46.1±9.8% in group RC at 2 and 4 weeks and 41.7±5.1% and 45.3±11.1% in group HA at 2 and 4 weeks. There was a tendency for larger amount of newly formed bone to exist at 2 weeks after surgery in group HA compared to other groups, although it did not reach statistical significance. There were significant differences between two time points in group NR. However, it did not show statistical significance in group RC and group HA. (Table 1, Fig.9)
IV. Discussion
Guided bone regeneration (GBR) procedure has been frequently performed due to the concept of contour augmentation in the esthetic zone and the efforts to minimize the complications related to bone deficiency around the endosseous implants. In GBR procedure, several types of barrier membranes are used with or without particulate bone grafts and/or bone substitutes3.
Non-resorbable expanded polytetrafluoroethylene (e-PTFE) membranes are biocompatible and highly effective on bone regeneration procedure having superior space maintaining and cell occlusive properties than resorbable membranes12. However, the need for removal surgery and high risk of bacterial infection when exposed leaded clinicians to prefer resorbable membranes13,14. Resorbable membranes are two categories of the natural products such as collagen or chitosan and the synthetic polyester-based membranes including PLA, PCL and PLGA15. Currently, synthetic membranes has been extensively researched because chitosan has been reported to have low blood compatibility and risk to disrupt the cell membrane15 and collagen membranes also have some limitations related with their origin, religious issue, mechanical properties, prices and so on7,8. However, these polyester-based membranes are mostly fabricated by melting or solvent casting16,17, which cannot help producing non-porous surface architectures less favorable to bone regeneration.
To overcome this surface non-porosity, electrospinning method was introduced since it can produce three dimensional porous network structure highly beneficial to cell attachment, proliferation, differentiation and new vascularization7,8. As electrospun polyester-based membranes do not have such good bone cell responses11,18, the techniques to intermix with microsized bioactive ceramic particles such as hydroxyapatite having osteoconductivity were tried soon thereafter19,20. However, intermixing different type of microsized structures was challenging because of their unmatching wettability, difficultly to achieve uniform dispersing and limitation of mixable ceramic particle quantity. Even though nanocomposites using sol-gel method provided a solution for these challenging problem21, these could not produce porous network structures needed for bone and tissue regeneration. Moreover, nanosized bioactive ceramic was too brittle to be maintained during the whole regeneration period11.
In the present study, three different types of membranes, or non-resorbable membranes, resorbable collagen membranes and HA-coated PCL/ CaO-SiO2 micro/nanofibrous membranes were utilized for GBR procedure on rabbit calvarial defects. HA coating onto PCL/ CaO-SiO2 microfibrous membranes was performed to give the membrane high osteoconductivity and was achieved by electrospinning of PCL and CaO-SiO2 gel solution followed by soaking within simulated body fluid for 7 days, which was confirmed by SEM investigation (Fig. 2A-2C).
PCL, one component of the membranes used in group HA, is slowly degradable synthetic polyester. The microsized PCL fibers can be served as adequate membrane scaffold since they have good mechanical properties such as high fracture-toughness and slow degradation rate which takes more than a year22. PCL has been approved by U.S. the Food and Drug Administration (FDA) in human clinical applications including a drug delivery carrier or suture material. The other component, or electrospun CaO-SiO2 nanofibrous structure has been reported to form HA on the surface of PCL fibrous scaffold in simulated body fluid, demonstrating their bioactivity and osteoconductivity11. It is known that both silicate ions and calcium ions released from CaO-SiO2 gel fibers play an important role during the process of HA forming mechanism. It is considered that the former is served as an initial nucleation site and the latter acts as an accelerator for HA crystal growth23.
Herein, it is suggested that following four factors should be satisfied to be served as optimal GBR membranes. First, membranes should have three dimensional porous network structure to provide space for bone regeneration and new vascularization24. In this study, this was obtained by electrospinning method in group HA. The sizes of these pores should be properly regulated because too large pores negatively affect cell occlusive property and also make the membrane weak and too small pores deteriorate tissue integration onto the membrane resulting in less bone formation25. In the present study, large amount of new bone matrices were formed all over the defect area, indicating that rapidly proliferating unwanted cells were effectively occluded and osteogenic cells and tissues were successfully integrated (Fig. 7, 8). Second, membranes are expected to have osteoconductivity which means they allow bone ingrowth by appositional bone formation when they are adjacent to the pristine bone. In group HA of this study, new bone formation was under way directly beneath the membrane or as closely incorporated pattern into the membrane structure demonstrating high osteoconductivity and vigorous bone formation (Fig. 7, 8). However, the osteoconductivity was confirmed in neither group NR (Fig. 3, 4) nor group RC (Fig. 5, 6). It can be explained that slowly resorbable PCL could be severed as adequate membrane scaffolds and favorable surface topography could be synergistic for tissue integration. Moreover, bioactive HA coating could promote the bone regeneration. Third, adequate mechanical properties are required to be maintained during the regeneration process. Microsized PCL is tough, ductile and strong enough to be served as a durable membrane11. Finally, membranes are recommended to have adequate biodegradability. In this study, biodegradable but long lasting PCL component and fast-degradable bioactive CaO-SiO2 component were well harmonized in PCL/ CaO-SiO2 microfibrous membranes.
The result of histomorphometric analysis shows a tendency for larger amount of newly formed bone to exist at 2 weeks after surgery in group HA (41.7±5.1%) compared to other groups (Group NR: 32.9±11.1%, Group RC: 32.5±15.8%), although it did not reach statistical significance (Table 1, Fig. 9). These results can be supportive that hydroxyapatite-coated membranes in group HA have promoting effect of new bone formation in the early stage of bone healing. As for intragroup analysis, there was significant difference between two time points in group NR only. This result are suggesting two possible explanations. One can be highly active new bone formation capacity of non-resorbable membranes throughout the healing time in group NR. The other can be large amount of new bone was already formed in the early healing time in group HA, which leaded not to reach statistical significant difference between two time points. The limitation of this study was small sample size and lack of bone volume comparison using 3-dimensional radiographic analysis.
Taken together, electrospun HA-coated PCL/ CaO-SiO2 micro/nanofibrous membranes fulfilled important factors required to be served as high-performance GBR membranes. For the clinical application, it is necessary to perform further experiments using bigger animal models and human clinical studies as well.
V. Conclusion
The resorbable hydroxyapatite-coated micro/nanofibrous membranes fabricated from PCL and bioactive CaO-SiO2 by concurrent electrospinning have high osteoconductivity and great effects on bone regeneration in GBR procedures especially in the early stage of bone healing, which was demonstrated by the experiments of rabbit calvarial defects in comparison with commercially available non-resorbable membranes and resorbable collagen membranes.
Moreover, these membranes have network-type porosity, osteoconductivity, adequate mechanical properties and biodegradability, so that they can be prospective and practical to be applied for clinical GBR procedures in the future.