I. Introduction
II. Material and Methods
1. Study subjects
2. Clinical procedure
3. Clinical examination
4. Statistical analysis
Ⅲ. Results
1. Implant survival rate
2. Marginal bone resorption
3. Probing depth
4. Plaque Index and calculus
5. Bleeding Index
6. Complications
Ⅳ. Discussion
V. Conclusion
I. Introduction
Among the zirconia-containing ceramics, tetragonal zirconia polycrystalline (Y-TZP) containing 3 mol% yttria has higher flexural strength ranged from 900 to 1,200 MPa. Because of its high flexural strength, Y-TZP is used in a variety of clinical applications; frameworks for all-ceramic posterior crowns, crown, bridge, implant abutments and dental implants, root canal posts1.
Most ZrO2 based-ceramic restorations have problems such opaqueness of the core, which should be masked with a translucent layer of veneer ceramic to achieve the natural appearance2. And monolithic zirconia is difficult to repair when a fracture occurs because the non-reactive surface of zirconia (acid-resistant ceramic) exhibit poor adhesion, a consistent problem of low bond strength to other substrates3. Another problem is the possibility of bone resorption around the implant by transferring more load to the implant because of its higher strength than other materials4-7.
Porcelain-fused-to-zirconia (PFZ) has been chosen to solve these problems8. Methods of veneering zirconia core include hand-layering technique with feldspathic porcelain, CAD-on technique and press-over technique. Recently, press-over technique for pressing lithium disilicate glass ceramic veneer has been introduced9. This technique is economical and simple because the lost wax press system can produce ceramic prostheses with or without computer aided design/computer aided manufacturing (CAD/CAM) system, and it can effectively fabricate the customized anatomical shape10.
Menini et al.11 reported that bone resorption and bone fracture can occur when pathological overload is present. Thus, it is important to control the forces transmitted on the bone-implant interface. According to an in vitro study, the force transmitted through the implant onto the peri-implant bone by zirconia (mean 641.8 N) was the greatest. Kim et al.9 reported that lithium disilicate pressed zirconia prostheses have similar fracture strength to monolithic zirconia and are clinically useful. Previous studies reported a comparison of survival rates and complications of zirconia and metal-ceramic prosthetic and all-ceramic1, 8. However, until now there are no clinical studies whether lithium disilicate pressed zirconia prostheses can solve strength and esthetic problems in posterior edentulous site compared to monolithic zirconia prostheses.
The aim of present study was to compare the clinical outcomes of lithium disilicate pressed zirconia prostheses and monolithic zirconia prostheses and to investigate the complications after 12 months of follow-up and to ascertain clinically enough to use lithium disilicate pressed zirconia prostheses.
II. Material and Methods
1. Study subjects
From 2015 to 2017, a total of 17 patients (male: 12, female: 5) were recruited at Pusan National University Dental Hospital (Table 1). This study was conducted with the consent of the patient under consideration of the National Bioethics Committee (IRB No. PNUDH- 2014-001-MD). 60 implant-supported prostheses were planned and followed up for 12 months after placement; 30 lithium disilicate pressed zirconia prostheses and 30 monolithic zirconia prostheses in the number of missing tooth. The subjects of this clinical study were patients over 21 years of age who were systemically healthy. Patients with missing tooth in the maxillary or mandibular posterior were included. They have sufficient bone width (at least 6 mm) and height (at least 10 mm) so that bone grafting is not necessary. Patients with one or more of the following diagnoses were excluded from subjects: alcohol or drug abuse, smoking, severe bruxism or other destructive habits, medical or psychological conditions preventing implant placement.
Table 1. Datum of patients and implants
Type of opposing dentition: N. Natural teeth, I. Implant prostheses.
Kyoung-Woo Roh et al. : Clinical Evaluation of Lithium Disilicate Pressed Zirconia and Monolithic Zirconia in Posterior Implant-supported Prostheses. Implantology 2019
2. Clinical procedure
External type implants (USII, Osstem Co., Seoul, Korea) were placed as the number of missing tooth. The surgical treatment was performed by specialists in periodontology, oral and maxillofacial surgery. Two-stage surgical procedure was performed according to the surgical instructions of the manufacturer. After completion of the second surgery, final impressions at fixture level were made with poly vinyl siloxane (Imprint II Garant regular/light body, 3M ESPE, Minnesota, USA) using the individual tray for open impression technique. The impressions of the opposing arch were made with alginate (aroma fine plus, GC, Tokyo, Japan). Titanium customized abutment (Pre-milled bar, Dio, Busan, Korea) that is set to the same margin level as gingiva were fabricated by CAD software (Exocad DentalCAD 2.2, Exocad, Darmstadt, Germany) and Milling machine (Trione Z, Dio, Busan, Korea). Provisional restorations were used to confirm the patient's discomfort for a month. A final impression was made at the abutment level for the final prostheses. The master casts were scanned by the AutoScan 3D Dental Scanner (Hangzhou Shining 3D Tech Co., Ltd., Hangzhou, China). The implant-supported prostheses are fabricated into two groups: the lithium disilicate pressed on zirconia-based prostheses (LP group, n=30) and the monolithic zirconia prostheses (MZ group, n=30). All the ceramic materials are presented in Table 2. To fabricate the LP group, 0.5 mm-thick zirconia coping (Zirtooth Fulluster, HASS, Gangneung, Korea) was made first. After fabricating the zirconia coping, liner powder (Rosetta Ceram Liner, HASS, Gangneung, Korea) was applied to the surface of the zirconia coping in order to improve the bond strength and wettability between the zirconia and the lithium disilicate glass ceramic veneer. And heat treatment was done. A wax veneer structure was fabricated by milling a wax block (TOTEM, Qingdao Totem Candle Industry, Shandong, China) using the Exocad software. The veneer was fixed to the coping by applying heat to its margin. And after investing, it was burned out at 880°C for 30 minutes (Burnout Furnace L 1/12, Nabertherm, Bremen, Germany). After that, glass ingot (Rosetta UltraPress, HASS, Gangneung, Korea) was put in to the investment ring, and the latter was pressed with the pressing furnace (Horizon Press, Shenpaz Dental Ltd., Migdal HaEmek, Israel) to bond the zirconia and the lithium disilicate glass ceramic. According to the instruction of the manufacturer, sandblasting (50 μm glass beads at 1 bar pressure) and glazing (IPS e.max Ceram glaze paste, Ivoclar Vivadent, Schaan, Leichtenstein) were performed. To fabricate the MZ group, a zirconia block (ZirtoothFulluster, HASS, gangneung, Korea) at green stage was milled. The occlusal surface is designed to have a uniform thickness of 1.5-2 mm. Milled zirconia prostheses was gone through the sintering and glazing. All implant-supported prostheses were cemented using self-adhesive resin cement (G-CEM LinkAce, GC America, Alsip, IL, USA). For the purpose of standardization, the same dental technician executed the manufacturing process.
Table 2. Materials used and properties of each group
Kyoung-Woo Roh et al. : Clinical Evaluation of Lithium Disilicate Pressed Zirconia and Monolithic Zirconia in Posterior Implant-supported Prostheses. Implantology 2019
3. Clinical examination
The following criteria were evaluated with reference to clinical examination and radiographs. Clinical examinations and radiographs were performed at 12 months after placement of the implant-supported prostheses, and complications were examined.
1) Implant survival rate
Implant survival rate was evaluated according to the criteria presented by Cochran et al.12. The evaluation criteria were: (1) no persistent discomfort such as pain, foreign body sensation, and abnormal sensation, (2) no persistent symptoms of peri-implant infection, such as pus discharge, and no relapse of such symptoms (3) no clinical mobility of the implants (4) no radiographic lucency around the implant, and no rapidly progressing bone loss.
2) Implant marginal bone resorption
Radiographs were taken using the parallel technique with a portable radiographic device (Port II, Genoray Co., Sungnam, Korea). The implant length and marginal bone level (distance from the implant platform to the top of the marginal bone) were measured using i-Solution (Olympus B × 51; Olympus Inc., Tokyo, Japan) and then the amount of marginal bone resorption compared with original bone level was calculated by comparing the implant length13.
3) Probing depth
The probing depth was measured at one point on each mesial, distal, buccal and lingual side around the implant in parallel with the long axis of the implant with Merritt-B periodontal probe, and then mean value was calculated14.
4) Plaque Index
According to the criteria of Mombelli et al.15, the plaque attached to the surface of the implant was measured and a score from 0 to 3 was assigned.
5) Bleeding Index
The bleeding tendency was assessed using a Merrit-B periodontal probe according to the criteria proposed by Mombelli et al.15
6) Calculus
Depending on the presence or absence of calculus, a score of 0 or 1 was given.
7) Complications
Complications were classified as (1) technically related, such as fracture of framework, connector fracture, chipping, crack, loss of retention, fixture fracture, screw fracture, screw loosening, (2) biologically related, such as peri-implantitis, crestal bone loss, gingival swelling, suppuration, food impaction. And frequency of complications was investigated.
4. Statistical analysis
To compare the marginal bone resorption and probing depth averages of the two groups, the independent T-test was used. Chi-square test was used to verify the significance of plaque index, calculus, bleeding index, and complications according to prostheses materials. All statistics were performed using SPSS (ver. 21.0, SPSS Inc., Chicago, IL, USA) at a significance level of 5%.
Ⅲ. Results
1. Implant survival rate
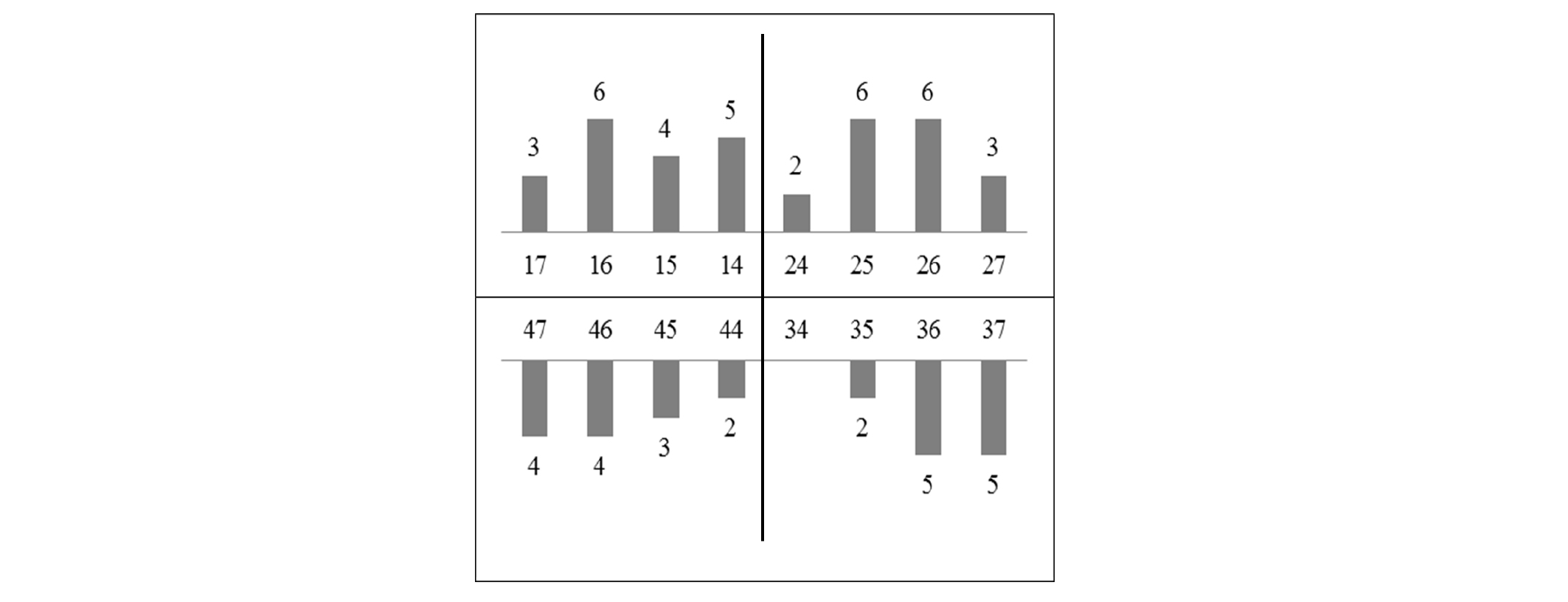
Of 17 patients with implant-supported prostheses, 60 implants were placed (Fig. 1): 30 implants in the LP group and 30 implants in the MZ group. All of 60 implants were loaded for 12 months after the definitive prostheses was placed. There were no failed implants and all implants were normal in function without clinical mobility (Table 3).
Table 3. Cumulative survival rate of the implants
| Follow-up periods (months) | LP group | MZ group | ||||
| Implants | Failed implants | CSR* (%) | Implants | Failed implants | CSR (%) | |
| 12 | 30 | - | 100 | 30 | - | 100 |
Kyoung-Woo Roh et al. : Clinical Evaluation of Lithium Disilicate Pressed Zirconia and Monolithic Zirconia in Posterior Implant-supported Prostheses. Implantology 2019
2. Marginal bone resorption
The mean values and standard deviations of implant marginal bone resorption at 1,3,12 months are shown in Table 4. The LP and MZ group showed 0.73±0.28 mm and 1.11±0.55 mm respectively at 12 months. The LP group had a significantly lower implant marginal bone resorption than the MZ group (p < 0.05).
3. Probing depth
The mean values and standard deviations of probing depth at 12 months are shown in Table 4. The LP group showed 3.15±0.45 mm and the MZ group showed 2.79±0.77 mm at 12 months. The LP group showed a significantly greater probing depth than the MZ group (p < 0.05).
Table 4. The average value of marginal bone resorption and probing dept h(12 months)
| LP group | MZ group | p | |||
| Mean | SD | Mean | SD | ||
| Marginal bone resorption (mm) | 0.73 | 0.28 | 1.11 | 0.55 | 0.02* |
| Probing depth (mm) | 3.153 | 0.45 | 2.79 | 0.77 | 0.032* |
Kyoung-Woo Roh et al. : Clinical Evaluation of Lithium Disilicate Pressed Zirconia and Monolithic Zirconia in Posterior Implant-supported Prostheses. Implantology 2019
4. Plaque Index and calculus
No plaque index was the most frequently observed in the MZ group (87%) and the score of 1 was the most frequently observed in the LP group (63%). The difference in the plaque index was significant (p < 0.05). The calculus in the LP group (47%) was significantly higher (p < 0.05) than in the MZ group (0%) (p < 0.05, Table 5).
5. Bleeding Index
No Bleeding index was the most frequently observed in the MZ group (70%). Relatively more bleeding was observed in the LP group and the difference in the bleeding index was significant (p < 0.05, Table 5).
Table 5. Plaque index and calculus, bleeding index
| Implant number | LP group† | MZ group† | p | |
| 30 | 30 | |||
| Plaque Index (%) | 0 | 7 | 87 | < 0.001* |
| 1 | 63 | 7 | ||
| 2 | 13 | 7 | ||
| 3 | 17 | - | ||
| Calculus (%) | 0 | 53 | 100 | < 0.001* |
| 1 | 47 | - | ||
| Bleeding Index (%) | 0 | 47 | 70 | 0.002* |
| 1 | 33 | 23 | ||
| 2 | 20 | 7 | ||
| 3 | - | - |
*Frequency distribution showed significant difference based on chi-square test (p< 0.05).
Kyoung-Woo Roh et al. : Clinical Evaluation of Lithium Disilicate Pressed Zirconia and Monolithic Zirconia in Posterior Implant-supported Prostheses. Implantology 2019
6. Complications
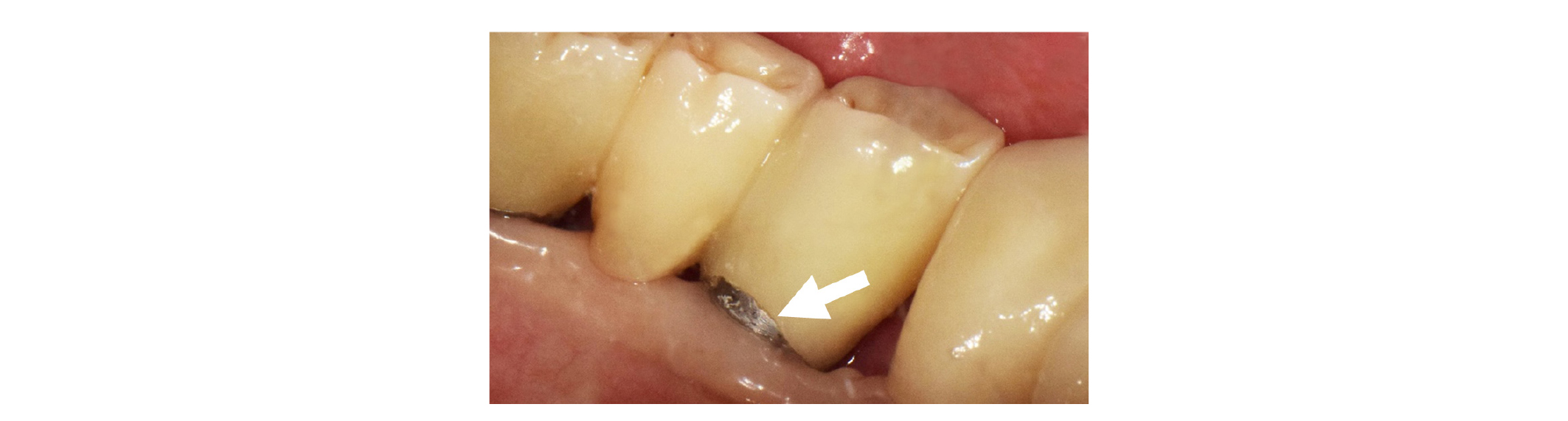
Complications were occurred in similar frequency in both groups. Chipping (3%) (Fig. 2) and food impaction (6%) were observed in the LP group, and connector fracture (3%) and more than 1.5 mm of bone loss (9%) were observed in the MZ group typically (Table 6).
Table 6. Type of clinical complications
| LP group | MZ group | |
| Connector fracture | - | 1 (3%) |
| Chipping | 1 (3%) | - |
| More than 1.5 mm of bone resorption | - | 3 (9%) |
| Food impaction | 2 (6%) | 1 (3%) |
| Total | 3 (9%) | 5 (15%) |
Ⅳ. Discussion
According to Albrektsson's implant success criteria, in this study, successful implantation was achieved by two groups showing under 1.5 mm bone resorption after one year of loading16. Many possible etiologies of early implant bone loss (from implant placement to 1-year post-loading) including surgical trauma, occlusal overload, peri-implantitis, the presence of microgap, reformation of biologic width, implant crest module, and others have been proposed. Among the various factors, occlusal overload is a major factor of implant failure17. Previous studies have indicated that occlusal overload often resulted in marginal bone loss or deosseointegration of successfully osseointegrated implants18-25. The crestal bone around the implant can be fulcrum for lever action when a bending moment is applied, which means that the implant may be more vulnerable to crest bone loss due to occlusal forces. Maria et al.11 reported that different restorative materials have a significantly impact on stress transfer to the peri-implant bone. Gracis et al.24 concluded that the stiffer the material, the higher the force transmitted onto the implant. According to Hooke’s law, the higher the modulus of elasticity of a material, the less the material will deform under pressure and the more likely the force will be transferred through the material. In this respect, zirconia transmits more force to the implant than other materials5-7.
Together with occlusal overload, peri-implantitis is main causative factors for implant failure. A correlation between plaque accumulation and progressive bone loss around implants has been reported in experimental and clinical studies1, 5, 21. Felicia et al.26 reported that zirconia has the lowest biofilm surface coating and biofilm thickness. These results suggest that zirconia prostheses are advantageous compared to lithium disilicate glass prostheses in aspect of bacterial adhesion. In other words, rough surfaces are more susceptible to peri-implantitis because bacterial plaques, also called bacterial plaques (biofilms), meet more favorable conditions on rough surfaces than on smooth surfaces. From the results of this clinical study, plaque index, calculus index, bleeding index, and probing depth were measured higher in LP group and marginal bone resorption was higher in MZ group. It is presumed that the period of 12 months was shorter until the inflammation process caused by plaque accumulation proceeded to peri-implantitis and bone resorption occurred. Rather, initial bone resorption is thought to be related to stress transmission. In fact, early crestal bone loss can provide a good environment for the growth of anaerobic bacteria. As a result, it may contribute more to subsequent bone destruction in following years. Nonetheless, the amount of crestal bone loss around the implant decreases after the first year of occlusal loading. Therefore, peri-implantitis may not be the primary causative factor in early implant bone loss17.
According to Larsson et al.27, porcelain veneer fractures more occurred in implant-supported zirconia prostheses than tooth-supported zirconia prostheses. Porcelain veneer fractures act like the periodontal ligament that absorbs shock. The results of this study showed that bone resorption was less in the LP group than in the MZ group for 12 months. The result is that the veneered lithium disilicate (96,000 MPa), which has a modulus of elasticity less than monolithic zirconia (210,000 MPa), absorbs the shock and transmits less stress to the cortical bone11.
The LP and MZ groups showed similar frequency of complications in this clinical study. LP group showed veneer chipping while the MZ group showed connector fracture (complete fracture). Among the complications, fractures are roughly classified into delamination, cohesive fracture within the veneer porcelain (chipping), cracks extending to the framework, and complete fracture28. Chipping or cracking limited to the porcelain veneer is the most commonly reported complication. To overcome chipping problem, over-pressing technique has been introduced. Over-pressing technique is a way to press specific ceramic onto the zirconia framework. However, chipping have been reported in zirconia-based prostheses veneered using the press-over technique29. One of the factors causing veneered zirconia-based restoration chipping is residual thermal stresses30. Residual stress can be caused by the mismatch of the larger coefficient of thermal expansion (CTE) of the veneering ceramic, thickness of the veneering porcelain, number of firing and the cooling rate. Therefore, slow cooling of the restoration above the glass transition temperature of the porcelain helps to reduce chipping31. It is also advantageous to use a lithium disilicate having a CTE (10.5-6/K-1) closer to zirconia (10.8-6/K-1) as build material to reduce CTE mismatch. Connector fracture was also observed in this study. So, when manufacturing multi-unit restorations, the area of the connector is also important for the durability and longevity of all-ceramic prostheses. A thinly designed connector area leads to critical strain faster than a thickly designed connector when the load is applied. In this study, the thickness was designed to be 10 mm2 for the mesial and 12 mm2 for the distal connector31.
Within the limitations of 1-year clinical pilot study, the periodontal index of lithium disilicate pressed zirconia was slightly worse, but the bone resorption was lower and only one chipping occurred. Therefore, lithium disilicate pressed zirconia is considered as a promising treatment option as much as monolithic zirconia in posterior implant-supported prostheses. And it is meaningful to be able to guess tendency in long-term follow-up. However, this study was not sufficient to demonstrate the clinical differences between lithium disilicate pressed zirconia and monolithic zirconia in posterior implant-supported prostheses due to short-term limitations. Because of the small size, including of single unit to four-unit prostheses is considered another limitation of this clinical study. Therefore, further studies should be conducted with larger sample sizes and over longer periods of time.
V. Conclusion
Within the limitations of the present study, the periodontal index of lithium disilicate pressed zirconia was slightly worse, but the bone resorption was lower and one chipping occurred. In other words, it is unreasonable to think that lithium disilicate pressed zirconia prostheses are absolutely superior or worse than monolithic zirconia prostheses within the limits of the study. Therefore, it can be considered as a treatment option for patients who require esthetics of their posterior implants, and who can manage their oral hygiene. In order to obtain more reliable clinical results between monolithic zirconia prostheses and lithium disilicate pressed zirconia, a larger number of subjects and comprehensive and long-term studies will be needed.